These Exploring Mixtures and Their Separation Ch 5 Notes are designed specifically for Class 9 students following the new book curriculum.
Why these notes are a must-have:
- NCERT Focused: Every point is mapped directly to the new NCERT book to ensure you don’t miss any scoring areas.
- Time-Saving: Complex separation methods like distillation and chromatography are simplified into easy-to-remember steps.
- Exam-Ready: Includes the exact definitions and diagrams often asked in Class 9 assessments.
- Use these Exploring Mixtures and Their Separation Ch 5 Notes to bridge the gap between reading the textbook and mastering the exam!
How Can We Classify Mixtures?
Classification of Mixtures
1. Homogeneous Mixture
(Solution)
- Has uniform composition throughout
- Every part looks and tastes the same
- Does not change with time — stays uniform
Examples:
| Mixture | Components |
|---|---|
| Sugar + Water | Sugar dissolved in water |
| Vinegar | Acetic acid in water |
| Soda | Carbon dioxide in water |
2. Heterogeneous
Mixture
- Has a non-uniform composition
- Components are visible and uneven
- Particles settle over time
Example:
| Mixture | Observation |
|---|---|
| Sand + Water | Sand visible, settles down |
| Oil + Water | Layers form, don’t mix |
Difference between
Homogeneous and Heterogeneous Mixtures
| Feature | Homogeneous | Heterogeneous |
|---|---|---|
| Composition | Uniform | Non-uniform |
| Visibility of parts | Not visible | Visible |
| Settling | No | Yes |
| Example | Sugar water | Sand + water |
Solutions
Basic Terms Related to Solutions
| Term | Meaning | Example |
|---|---|---|
| Solute | Substance that dissolves | Sugar |
| Solvent | Substance that dissolves the solute | Water |
| Solution | Solute + Solvent mixed | Sugar water |
Concentration of a Solution
- Amount of solute dissolved in a given amount of solvent or solution
- The right proportion is always essential
- Too little or too much solute changes the solution’s purpose
Real-life examples:
- ORS — fixed salt + sugar in fixed water
- Pesticides — too little = ineffective, too much = damages crops/environment
Ways to Express Concentration
1. Mass by Mass percentage (% m/m or % w/w)
Formula:Mass by Mass percentage = (Mass of solute ÷ Mass of solution) × 100
► Used for homogeneous & heterogeneous mixtures
Examples: Milk powder, spice mixes, packaged food labels (salt, sugar, protein content)
2. Mass by Volume percentage (% m/v or % w/v)
Formula: Mass by Volume percentage = (Mass of solute ÷ Volume of solution) × 100
► Used when volume is easier to measure than mass
Examples: Medicines, lab solutions, 5% glucose solution
3. Volume by Volume percentage (% v/v)
Formula: Volume by Volume percentage= (Volume of solute ÷ Volume of solution) × 100
► Used when two liquids are mixed (miscible liquids)
Examples: Perfumes, cosmetics, vinegar
In short,
| Type | Solute Unit | Solution Unit | Used For |
|---|---|---|---|
| % m/m | grams | grams | Food, powders |
| % m/v | grams | millilitres | Medicines, labs |
| % v/v | millilitres | millilitres | Liquids, perfumes |
Solubility
- Solubility = the maximum amount of solute that dissolves in 100 mL or 100 g of solvent at a given temperature
- Saturated Solution = cannot dissolve any more solute at that temperature
Effect of Temperature
| Type | Effect of ↑ Temperature |
|---|---|
| Solid in liquid | Solubility increases |
| Gas in liquid | Solubility decreases |
● A graph of solubility vs temperature = Solubility Curve
● Solubility is used to separate substances from mixtures
Solubility Curve
It tells how much solute dissolves in water at different temperatures.
A solubility curve is a graph that shows:
- X-axis → Temperature (°C).
- Y-axis → Solubility (g per 100 g water).
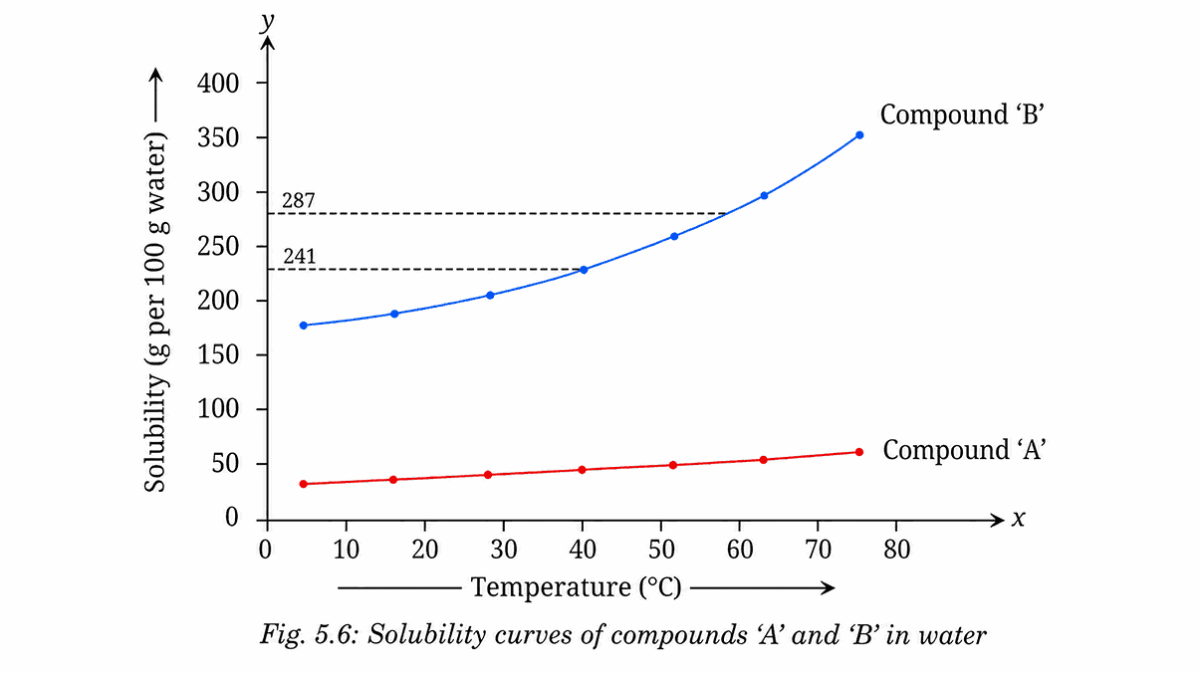
Ans: Activity 5.2: Let us represent solubility graphically:
(i) The solubility of compound ‘A’ at 20°C is
less than its solubility at 60°C
(ii) The solubility of compound ‘B’ at 20°C is
less than its solubility at 60°C
(iii) The solubility of
Compound A increases more than that of Compound B
Methods of Separation of Homogeneous Mixtures
1. Crystallization
What is it?
●Process of forming pure crystals from a saturated solution
●Based on the principle that solubility changes with temperature
How it works
●Prepare a saturated solution at high temperature
●Cool it gradually → excess solute can no longer stay dissolved → separates as crystals
Example:
287g of compound B dissolves at 60°C, but only 241g stays dissolved at 40°C → rest separates as crystals
What is a Crystal?
A solid with particles arranged in a regular geometric pattern
Natural Examples:
| Crystal | How it Forms |
|---|---|
| Rock salt | Natural mineral deposits |
| Mishri (candy sugar) | Cooling of sugar solution |
| Snowflakes | Water vapour freezes in the air |
| Frost on windows | Water vapour turns to ice crystals |
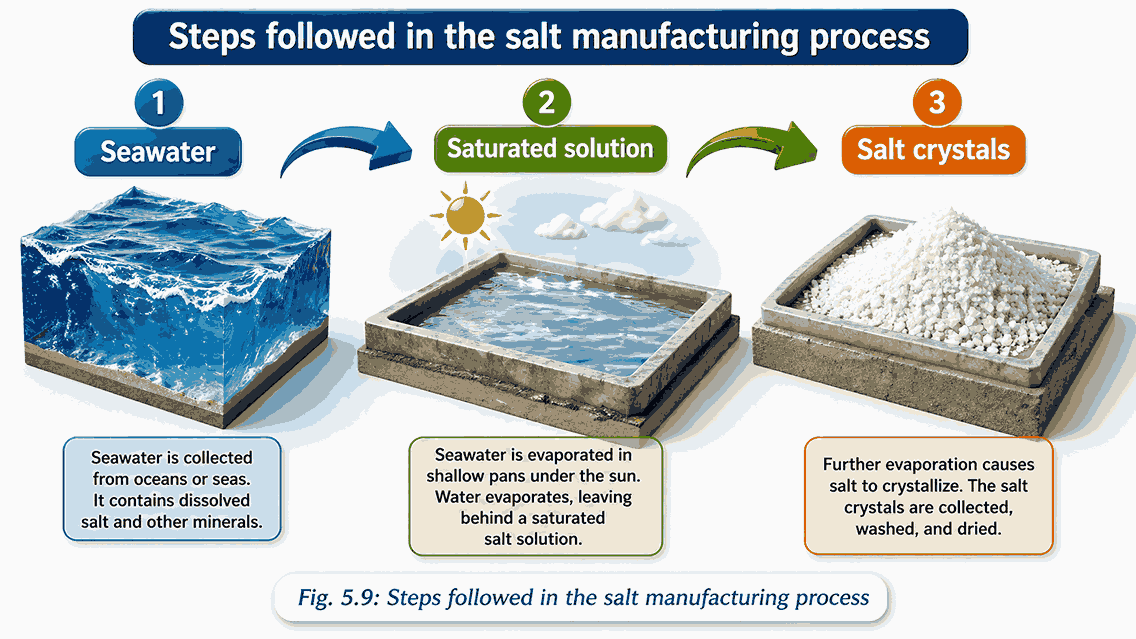
Uses of Crystallization
● Separate two solids soluble in the same solvent
● Purify solids by removing impurities
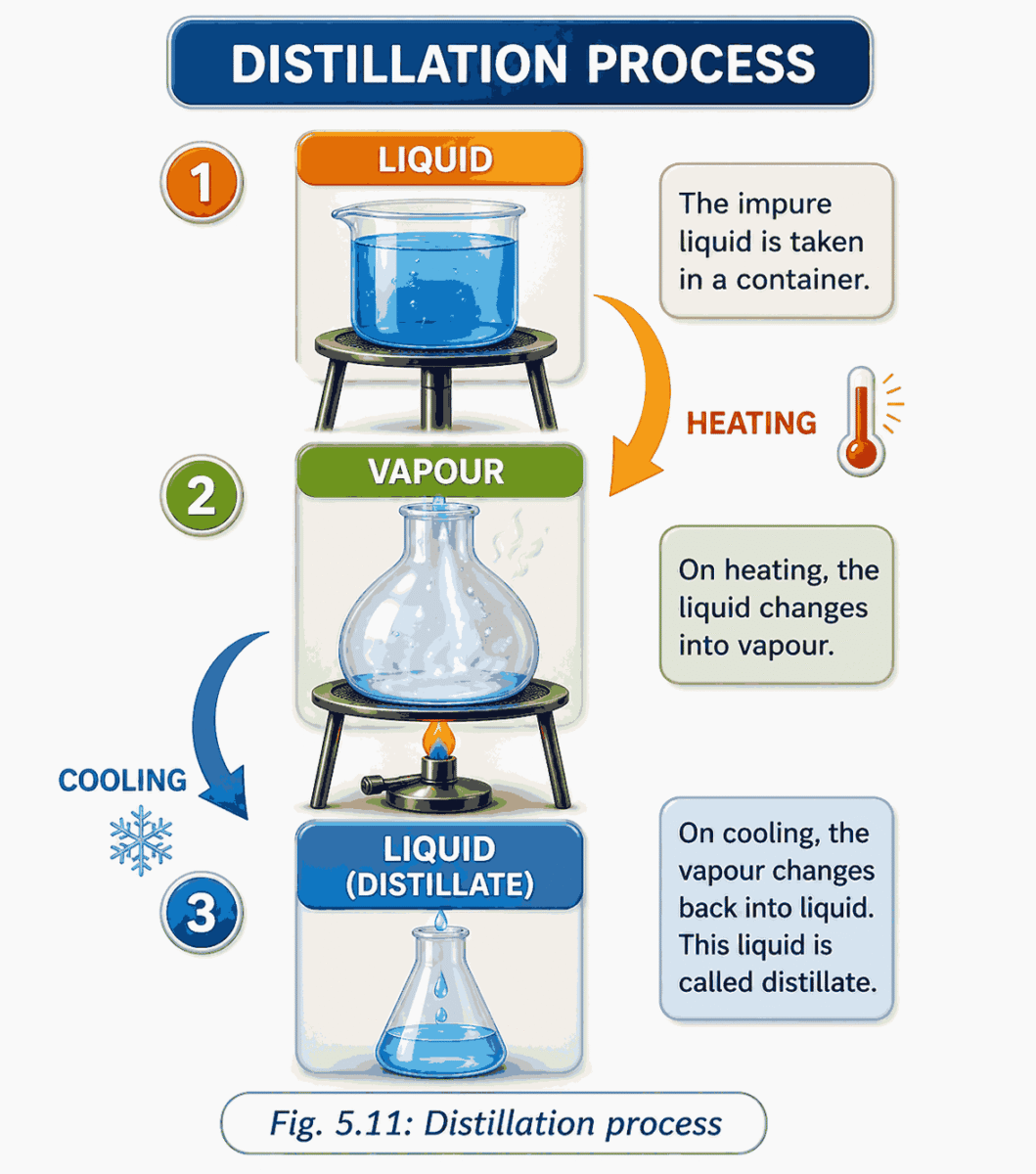
2. Distillation
What is it?
● Separation of two miscible liquids (or a liquid from a dissolved solid) by using the difference in boiling points
How it works
● Heat the mixture
● Liquid with a lower boiling point vaporises first
● Vapour passes through a condenser (cooled by water/air)
● Vapour turns back to pure liquid
● Collected in a separate vessel
Condition for distillation
The boiling points of the two liquids must differ by at least ~25°C
Example:
| Liquid | Boiling Point |
|---|---|
| Acetone | 56°C |
| Water | 100°C |
| Difference | ~44°C ✅ |
Uses of
Distillation
- Separate miscible liquids (e.g., acetone + water)
- Recover the solvent from a solution
- Separate liquid from dissolved solids
3. Paper Chromatography
What is it?
● Method to separate components of a mixture using differences in how they interact with paper and solvent
How it works
● A solvent moves up the paper
● Carries different components at different speeds
● Components separate based on their speed of travel
Uses of Paper Chromatography
| Application | What gets Separated |
|---|---|
| Black ink on wet paper | Different colour pigments |
| Spinach leaf extract | Green pigments |
| Flower petals | Coloured pigments |
●Water works as a solvent in some cases
●For others, alcohol or mixed solvents may be needed
Quick Comparison of Methods
| Method | Mixture Type | Principle | What is Recovered |
|---|---|---|---|
| Crystallization | Solid in liquid | Solubility vs Temperature | Pure solid crystals |
| Distillation | Liquid in liquid / solid in liquid | Difference in boiling points | Pure liquid(s) |
| Paper Chromatography | Mixed components | Interaction with paper & solvent | Liquid in liquid/solid in liquid |
How Can We Separate the Components of Heterogeneous Mixtures?
1. Separation of two immiscible liquids
We use a separating funnel
Separating Mustard Oil from Water
- Method: Separating funnel (5 mL oil + 20 mL water)
- Observation: Two layers form—oil (upper, yellow), water (lower)
- Reason: Oil is less dense than water; immiscible
- Steps:
- Drain the lower water layer via the stopcock
- Discard the small mixed portion at the interface
- Collect the oil layer separately
Types of Heterogeneous Mixtures
| Type | Examples |
|---|---|
| Liquid-Liquid (immiscible) | Oil + Water |
| Solid-Liquid | Sand + Water, Iron + Sulfur |
| Gas-based | Smoke, Fog, Dust in air |
| Solid-Solid | Iron filings + Sawdust |
Most gas mixtures are homogeneous (e.g., H₂ + O₂ in rocket fuel). Most solid-solid mixtures are heterogeneous
2. Sublimation
What is it?
● Solid converts directly to vapour (without becoming liquid) on heating,
● Vapour converts directly back to a solid on cooling = deposition.
How it Separates
● Only one component sublimes; the other stays behind
● E.g., Camphor sublimes → Sand does not → They separate
Examples of Sublimable Substances
| Substance | Use/Note |
|---|---|
| Camphor | Common lab example |
| Naphthalene | Can replace camphor |
| Dry ice (solid CO₂) | Used in ice cream storage |
3. Alloys
What is an Alloy?
A homogeneous mixture of two or more metals, or a metal + non-metal
Formed by melting metals together at high temperatures
Cannot be separated by physical methods
Why are Alloys Made?
Stronger, more rigid, and more corrosion-resistant than pure metals
Common Alloys
| Alloy | Main Components | Approximate Composition |
|---|---|---|
| Brass | Copper + Zinc | ~80% Cu, ~20% Zn |
| Bronze | Copper + Tin | ~80% Cu, ~20% Sn |
| Stainless Steel | Iron + Carbon + Chromium + Nickel + Molybdenum | C: 0.03–0.8%, Cr: 16–18%, Ni: 10–14%, Mo: 2–3% |
4. Suspensions
What is it?
● A heterogeneous mixture where solid particles do not dissolve but remain suspended in a medium
● Particles are visible to the naked eye
● Particles are larger than those in a solution
Examples
● Sand in water
● Sawdust in water
● Tea leaves in water
● Muddy water
Separating Suspensions [Muddy Water]
| Steps | Method | Limitation |
|---|---|---|
| 1st | Leave undisturbed → heavier particles settle | Water may still be cloudy |
| 2nd | Filtration (cloth/filter paper) | Removes larger particles only |
| 3rd | Centrifugation and/or Coagulation | Used when filtration is not enough |
A. Centrifugation
What is it?
● Spinning a mixture in a tube at high speed
● Uses centrifugal force (outward force on a body in circular motion)
How it Works
● Tube spins → becomes horizontal
● Heavier particles move outward → settle at the bottom
● Lighter liquid stays at the top
Uses of Centrifugation
| Application | What gets Separated |
|---|---|
| Laboratories | Red blood cells + Plasma (from blood) |
| Chemical industries | What gets separated |
B. Coagulation
What is it?
● A process where fine suspended particles clump together to form larger particles
● The substance causing this is called a coagulant
How it Works
● Add coagulant (e.g., alum) to muddy water
● Fine particles clump together
● Larger clumps settle by gravity (sedimentation)
● Clear water separated by decantation or filtration
| Example | Coagulant Used |
|---|---|
| Muddy water purification | Alum (fitkari) |
| Cheese (paneer) from milk | Lemon juice or Vinegar (acid) |
In cheese-making, the acid causes milk proteins to coagulate and form paneer
5. Colloids
What is a Colloid?
- A mixture that is neither a solution nor a suspension
- Particles are uniformly dispersed, but larger than in a solution
- Particles do not settle over time (unlike suspensions)
Examples
Blood, Milk, Tomato sauce, Ice cream
Solution vs Colloid vs Suspension
| Feature | Solution | Colloid | Suspension |
|---|---|---|---|
| Particle Size | < 1 nm | 1–1000 nm | > 1000 nm |
| Visible to naked eye | ❌ No | ❌ No | ✅ Yes |
| Settles over time | ❌ No | ❌ No | ✅ Yes |
| Examples | Sugar water | Blood, Milk | Sand + Water |
| Separation method | Crystallization/Distillation | Centrifugation | Filtration/Sedimentation |
Why is Blood a Colloid?
| Observation | Conclusion |
|---|---|
| Blood cells not visible to naked eye | ❌ Not a suspension |
| Blood can be centrifuged & coagulates | ❌ Not a solution |
| Particles don’t settle, uniformly dispersed | Blood cells not visible to the naked eye |
Tyndall Effect
What is it?
- Scattering of light by particles in a colloid or suspension
- Named after the scientist John Tyndall
When does it occur?
| Mixture | Light Path Visible? |
|---|---|
| Solution (transparent) | ❌ No |
| Colloid | ✅ Yes |
| Suspension | ✅ Yes |
Real-life Examples
- Light entering a dark room through a small hole (dust scatters light)
- Floodlights in a sports stadium
- Beam of light through smoke or fog
Dispersed Phase & Dispersion Medium
| Term | Meaning | Equivalent to |
|---|---|---|
| Dispersed Phase | Solute-like scattered particles | Solute |
| Dispersion Medium | Medium in which particles are suspended | Solvent |
Separation in Nature & Everyday Life
- Kidneys — remove waste from blood
- Sewage treatment — sedimentation → coagulation → filtration → reuse
- Waste segregation at home:
- Dry waste (plastic, paper, glass, metal) → Recycled
- Wet waste (food scraps, peels) → Composted
- Lithium recovery from old phone/laptop batteries → research ongoing
FAQs: Exploring Mixtures and Their Separation Ch 5 Notes
How many times should I revise these Ch 5 notes for exams?
For the best results, follow the 3-Pass Rule:
First Pass: Read the notes immediately after finishing the NCERT textbook chapter to solidify concepts.
Second Pass: Review them 3 days later to move the info into long-term memory.
Third Pass: A final “speed-read” 24 hours before your exam to keep definitions fresh.
How do I use these notes to prepare for Class 9 Science practicals?
These notes include step-by-step logic for methods like filtration, crystallization, and centrifugation. Before your lab session, read the “How it Works” section for each method in these notes so you can explain the science behind your experiment to the examiner.
What is the most important topic in Exploring Mixtures and Their Separation?
While the whole chapter is vital, examiners frequently focus on the differences between solutions, colloids, and suspensions, as well as the Tyndall Effect. Use the comparison tables in these notes to ensure you can list at least three distinct differences for each.
Can I rely solely on these notes for my final exams?
These notes are an indispensable revision tool designed to save you time. However, you should always read the NCERT textbook at least once to understand the full context. Use these notes to capture the “meat” of the chapter—the formulas, key definitions, and separation principles—during your final countdown
How should I practice the numerical problems in this chapter?
Focus on the Concentration of a Solution section in the notes. Memorize the three formulas (Mass by Mass, Mass by Volume, and Volume by Volume). Practice at least two examples for each formula from your textbook, then use the notes to quickly cross-check that you’ve used the correct units (grams vs. milliliters).



![[New Book] How Forces Affect Motion Notes Ch 6](https://studyless.in/wp-content/uploads/2026/04/How-Forces-Affect-Motion-Notes-Ch-6-2-1200x675.jpg)
