After you have prepared the chapter 5 Exploring Mixtures and their Separation Class 9 NCERT, now its time to check your preparation.
“Answers Ch 5 Exploring Mixtures and their Separation” is created to give you the best and to the point answers to all the questions of Revise, Reflect, Refine at the end of the chapter.
The answers to the end of the chapter questions are strictly on the basis of the chapter 5 of science textbook, Exploration for class 9.
Question 1
Which of the following mixtures are correctly classified as homogeneous (Hm) and heterogeneous (Ht)? Choose the correct
option.
(i) Air — Hm, Milk — Ht, Sugar solution — Hm, Smoke — Hm
(ii) Brass — Ht, Fog — Ht, Vinegar — Ht, Muddy water — Hm
(iii) Copper sulfate solution — Hm, Salt solution — Hm, Milk — Hm,
Bronze — Hm
(iv) Muddy water — Ht, Milk — Ht, Blood — Ht, Brass — Hm
Answer
Option (iv) is correct.
- Muddy water — Ht ✓ (sand particles are visible and settle — heterogeneous)
- Milk — Ht ✓ (milk is a colloid; fat droplets are dispersed — heterogeneous)
- Blood — Ht ✓ (blood is a colloid with cells dispersed in plasma — heterogeneous)
- Brass — Hm ✓ (brass is an alloy of copper and zinc, uniform throughout — homogeneous)
Why the others are wrong:
- Option (i): Smoke is heterogeneous (solid particles in gas), not Hm.
- Option (ii): Brass is homogeneous, not Ht. Vinegar is homogeneous, not Ht.
- Option (iii): Milk is heterogeneous (colloid), not Hm.
Question 2
Choose the correct options, and explain the reason for the correct and incorrect options. Which among the following mixtures shows the Tyndall Effect?
A mixture of:
(a) air and dust particles
(b) copper sulfate and water
(c) starch and water
(d) acetone and water
(i) a and b (ii) b and d (iii) a and c (iv) c and d
Answer
Option (iii) — a and c — is correct.
The Tyndall effect occurs when particles scatter light. This happens only in colloids and suspensions, not in true solutions.
- (a) Air and dust particles ✓ — Dust particles are large enough to scatter light. The visible beam of light in a dusty room is a perfect example of the Tyndall effect.
- (c) Starch and water ✓ — Starch forms a colloid in water. The starch particles (1–1000 nm) scatter light and show the Tyndall effect.
- (b) Copper sulfate and water ✗ — This is a true solution. Particles are smaller than 1 nm and cannot scatter light. No Tyndall effect.
- (d) Acetone and water ✗ — This is also a true solution (both are miscible liquids). No Tyndall effect.
Question 3
A mixture can be categorised as a solution, a suspension, or a colloid, each possessing distinct properties. Utilise the words or phrases provided in the box to fill in Table 5.2. Words and phrases may be used more than once.
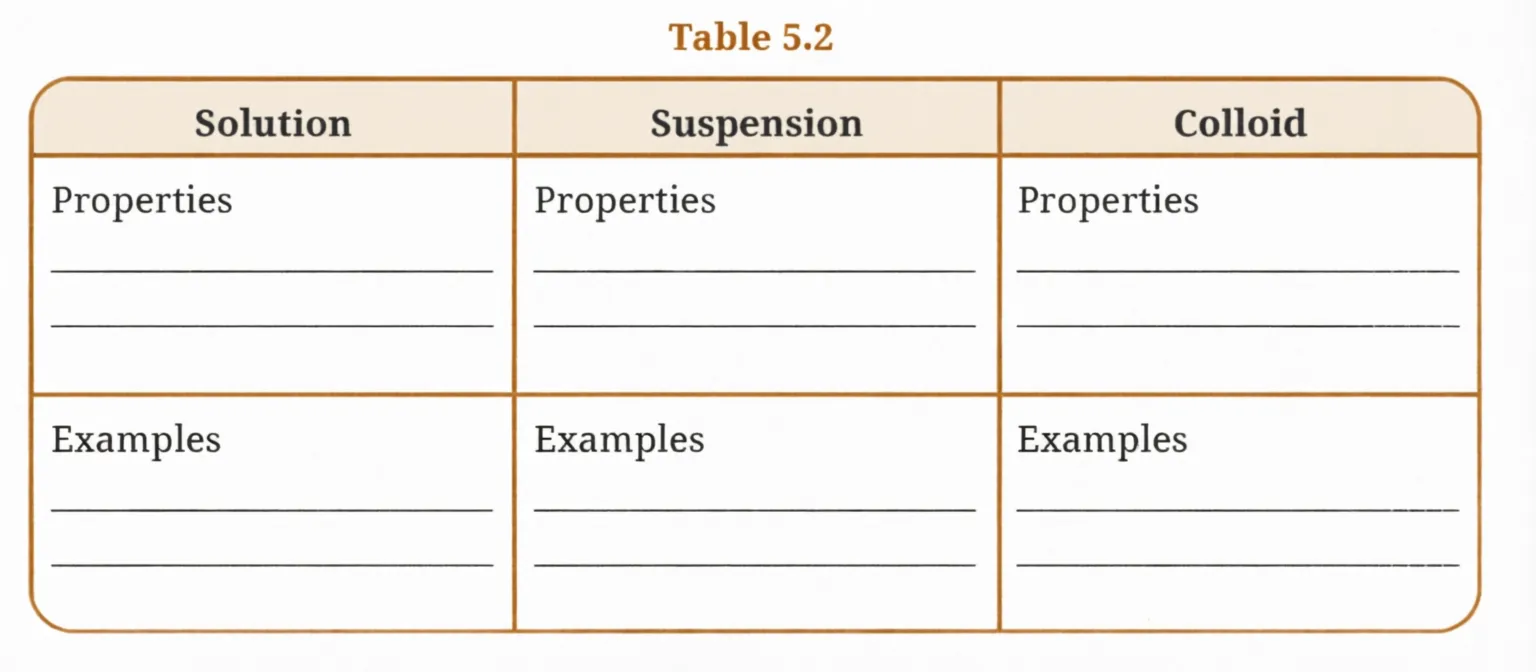
Answer
Table 5.2 — Completed
| Solution | Suspension | Colloid | |
|---|---|---|---|
| Properties | Small-sized particles (less than 1 nm diameter) | Large-sized particles (more than 1000 nm in diameter) | Moderate-sized particles (1–1000 nm) |
| Particles remain evenly distributed | Settles down when left undisturbed | Particles remain evenly distributed | |
| Transparent | Heterogeneous mixture | Scatters light (Tyndall effect) | |
| Cannot be separated by filtration | Separates by filtration | Cannot be separated by filtration | |
| Does not settle down | — | Does not settle down | |
| Examples | Salt solution, Brass | Sand in water, Mud | Small-sized particles (less than 1 nm in diameter) |
Question 4
Solve the following problems:
(i) A cake recipe uses dry ingredients, namely, 75 g of sugar for 420 g of all-purpose flour and 5 g of sodium hydrogencarbonate.
Express the concentration of each component in the mixture using an appropriate method.
(ii) A brass alloy contains 70% copper by mass. Calculate the quantities of copper and zinc present in 120 g of brass.
Answer
(i) A cake recipe uses dry ingredients, namely 75 g of sugar for 420 g of all-purpose flour and 5 g of sodium hydrogencarbonate. Express the concentration of each component in the mixture using an appropriate method.
Answer:
Since all components are solids being mixed together, mass by mass percentage (% m/m) is the most appropriate method here.
Total mass of dry mixture = 75 + 420 + 5 = 500 g
Concentration of sugar: % m/m = (Mass of sugar / Total mass) × 100 = (75 / 500) × 100 = 15% m/m
Concentration of flour: % m/m = (420 / 500) × 100 = 84% m/m
Concentration of sodium hydrogencarbonate: % m/m = (5 / 500) × 100 = 1% m/m
(ii) A brass alloy contains 70% copper by mass. Calculate the quantities of copper and zinc present in 120 g of brass.
Answer:
Mass of copper = 70% of 120 g = (70/100) × 120 = 84 g
Since brass is a mixture of only copper and zinc:
Mass of zinc = 120 − 84 = 36 g
So, in 120 g of brass: Copper = 84 g, Zinc = 36 g.
Question 5
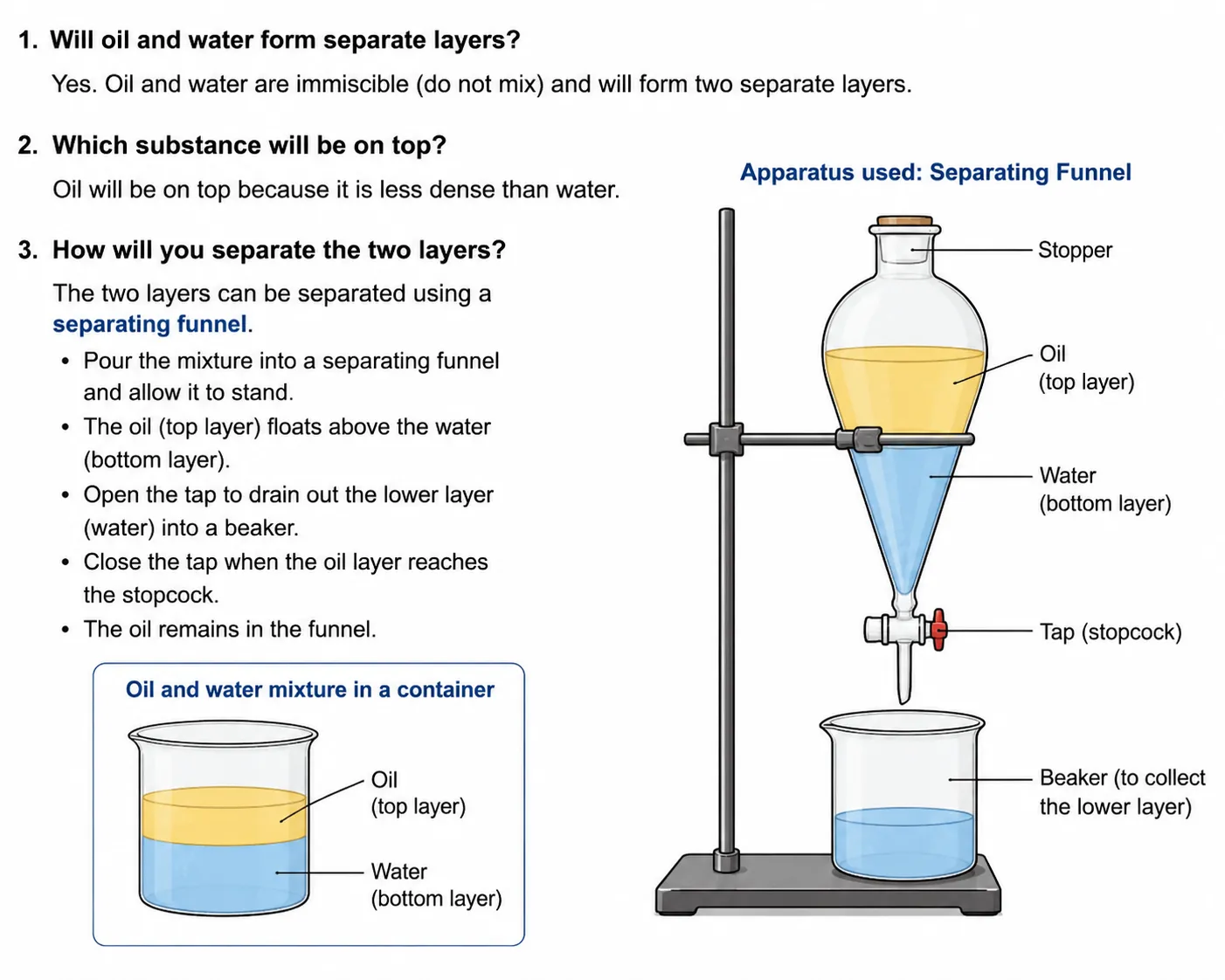
The label on a cooking oil pack says one litre (910 g). If this oil is mixed with water, will it form a separate layer? If so, which substance will be on top? How will you separate the two layers? Also, draw the diagram of the apparatus used.
Answer
Yes, oil and water will form two separate layers. Oil and water are immiscible — they do not dissolve in each other.
Oil will be on top. This is because oil (density ≈ 0.91 g/mL) is less dense than water (density = 1 g/mL). The less dense liquid always floats above the denser one.
Separation method: Separating Funnel
The mixture is poured into a separating funnel. The two layers settle — water at the bottom and oil on top. The stopcock is opened slowly to drain the lower layer (water) into a beaker. The stopcock is closed just before all the water drains. The oil layer is then collected separately.
Diagram of Separating Funnel:
Question 6
Assertion (A): Solutions do not exhibit the Tyndall effect.
Reason (R): The particles in solutions are larger than 100 nm, so they cannot scatter light.
Choose the correct option:
(i) Both A and R are true, and R is the correct explanation of A.
(ii) Both A and R are true, but R is not the correct explanation of A.
(iii) A is true, but R is false.
(iv) A is false, but R is true.
Answer
Answer: Option (iii) — A is true, but R is false.
Assertion is TRUE: Solutions do not show the Tyndall effect because their particles are too small to scatter light.
Reason is FALSE: The particles in solutions are smaller than 1 nm (not larger than 100 nm). Because the particles are so tiny, they cannot scatter light. The Reason states the opposite of the truth, so it is incorrect.
Question 7
How would you separate the mixtures given in Table 5.3? Mention the reason for choosing your method. If a mixture cannot be separated, explain why.
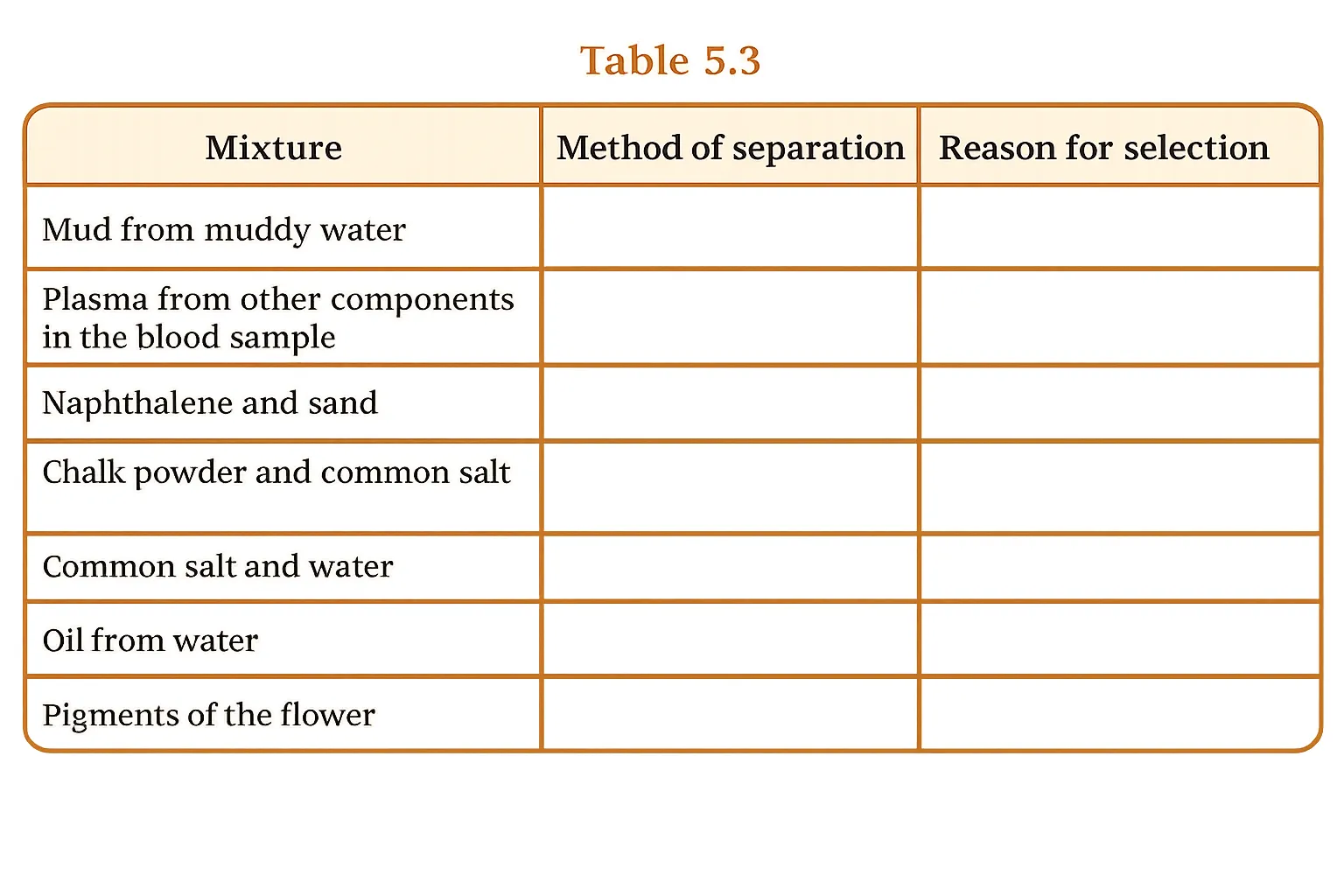
Answer
Answer:
| Mixture | Method of Separation | Reason for Selection |
|---|---|---|
| Mud from muddy water | Coagulation + Filtration / Centrifugation | Mud particles are large and suspended. Alum (coagulant) clumps them together for easier settling and filtration. Centrifugation works for smaller suspended particles. |
| Plasma from other components in blood | Centrifugation | Blood is a colloid. Spinning at high speed separates heavier red blood cells and platelets from lighter plasma. |
| Naphthalene and sand | Sublimation | Naphthalene sublimes on gentle heating (turns directly to vapour) while sand remains behind. The vapour is collected on a cool inverted funnel and deposits as solid naphthalene. |
| Chalk powder and common salt | Dissolve in water + Filtration + Evaporation | Chalk does not dissolve in water; salt does. Filter to remove chalk, then evaporate the filtrate to recover salt. |
| Common salt and water | Evaporation / Distillation | Evaporation removes water to collect salt. Distillation is used if both salt and pure water are needed. |
| Oil from water | Separating Funnel | Oil and water are immiscible and form two layers due to density difference. A separating funnel separates them easily. |
| Pigments of the flower | Paper Chromatography | Oil and water are immiscible and form two layers due to the density difference. A separating funnel separates them easily. |
Question 8
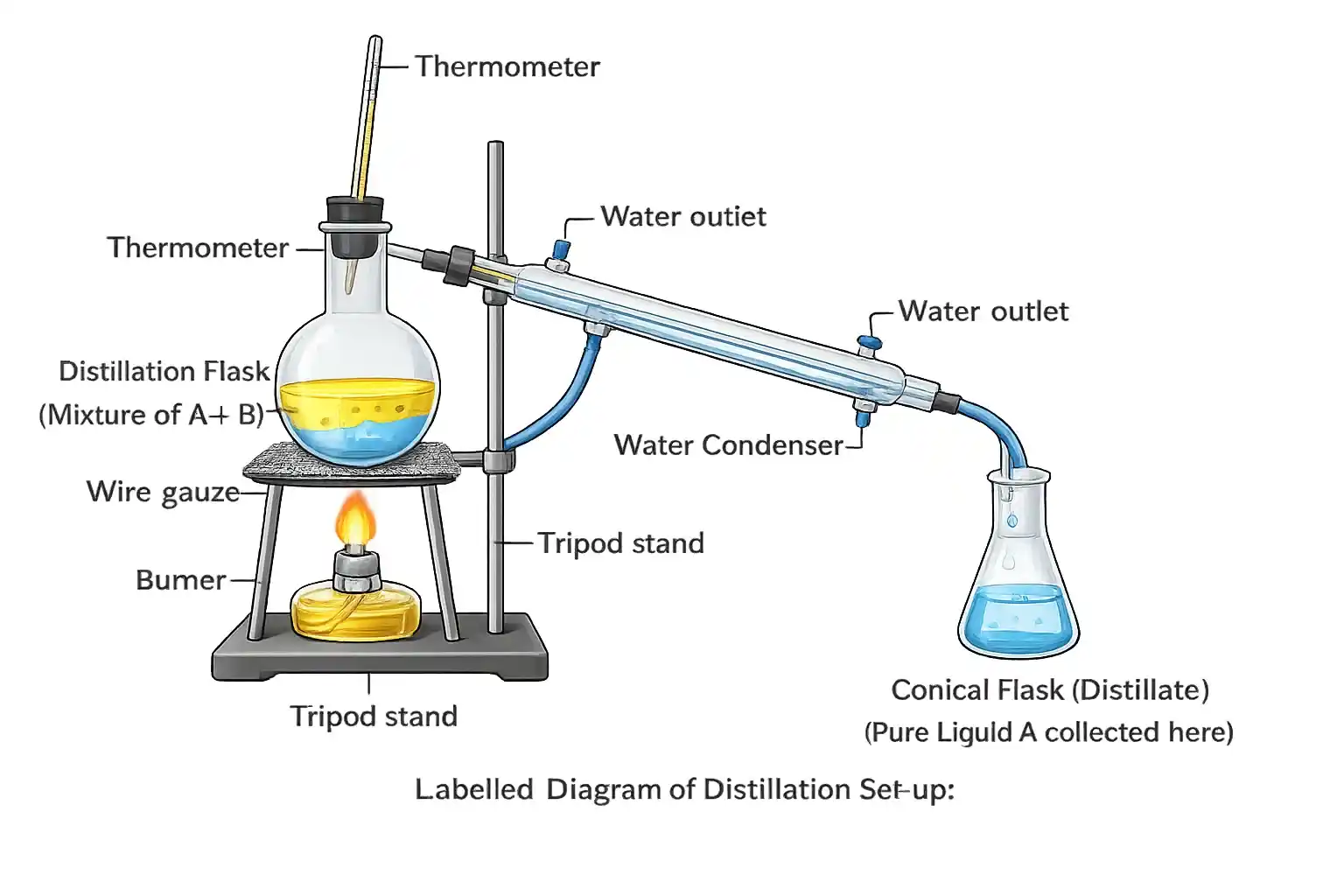
Two miscible liquids, A and B, are present in a mixture. The boiling point of A is 60 °C and the boiling point of B is 90 °C. Suggest a method to separate them. Also, draw a labelled diagram of the method suggested.
Answer
Answer:
Distillation is the best method to separate liquids A and B.
The difference in their boiling points is 90 − 60 = 30°C, which is greater than the minimum required difference of 25°C. This makes distillation effective.
How it works: The mixture is heated in a distillation flask. Liquid A (boiling point 60°C) vaporises first. The vapour passes through the condenser, cools down, and is collected as pure liquid A in the conical flask. Liquid B (boiling point 90°C) remains in the distillation flask until all of A is separated, then B is collected similarly.
Question 9
Compare evaporation, crystallisation, and distillation. In which situation would you prefer each of these over the others?
Answer
Answer:
| Feature | Evaporation | Crystallization | Distillation |
|---|---|---|---|
| What it does | Removes solvent by heating; solute is collected | Removes solvent by heating; the solute is collected | Separates miscible liquids or recovers solvent from a solution |
| Solvent recovered? | No | No | Yes |
| Product obtained | Solid solute | Pure crystals of solute | Pure liquid (distillate) |
| Best used when | You only need the solid solute and don’t need the solvent | You need a pure solid and the mixture has impurities | You need a pure solid, and the mixture has impurities |
When to prefer each:
Use Evaporation when you want to quickly recover a solid (like salt) from a solution and do not need the water/solvent.
Use Crystallization when you need a pure solid with a well-defined shape and structure (like copper sulfate crystals), especially when impurities are present. Slow cooling gives large, pure crystals.
Use Distillation when you need to recover the solvent as well (like getting pure water from saltwater), or when you need to separate two miscible liquids like acetone and water.
Question 10
Blood is an example of a colloidal mixture. (i) What would happen if blood behaved like a true suspension inside the body? (ii) In a blood sample, identify the dispersed phase and the dispersion medium
Answer
Answer:
(i) If blood behaved like a true suspension:
- Blood cells (larger than 1000 nm) would settle to the bottom of blood vessels over time, just like mud settles in muddy water.
- Blood would not flow smoothly — it would clog vessels.
- Oxygen, nutrients and hormones could not be transported effectively.
- The body’s organs would fail to receive what they need to function.
- Life as we know it would not be possible.
(ii) Dispersed phase and dispersion medium in blood:
- Dispersed phase = Blood cells (red blood cells, white blood cells, platelets) — these are the solid particles dispersed throughout.
- Dispersion medium = Plasma — this is the liquid in which the blood cells are suspended.
Question 11
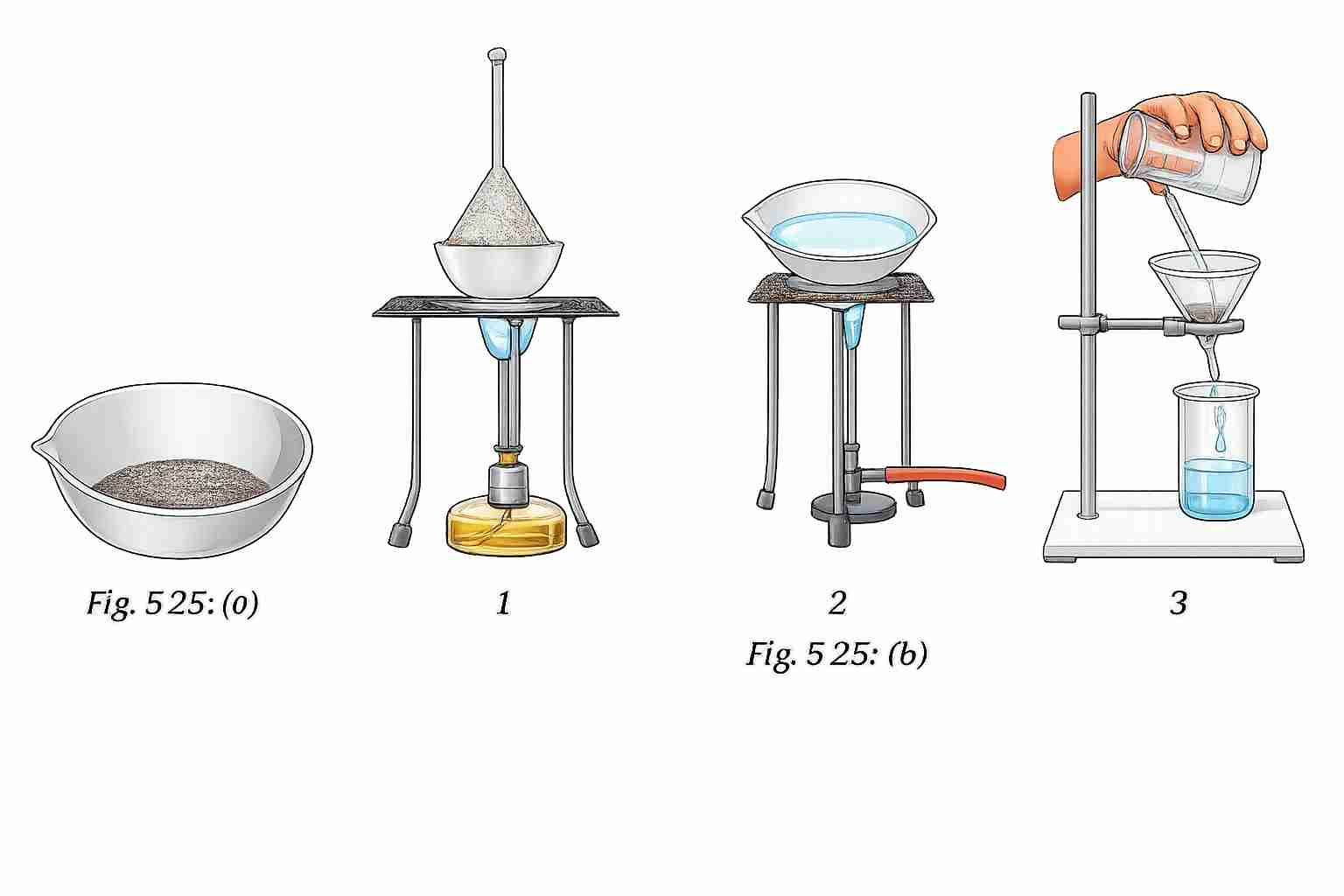
You are given a mixture of sand, common salt and naphthalene (Fig. 5.25a). The Fig. 5.25b depicts various steps used to separate the
components of this mixture. Identify and write down the correct sequence of separation techniques.
Answer
Answer:
The correct sequence is:
Step 1 → Sublimation → Step 2 → Dissolving in water + Filtration → Step 3 → Evaporation
Flowchart of the Separation Process:
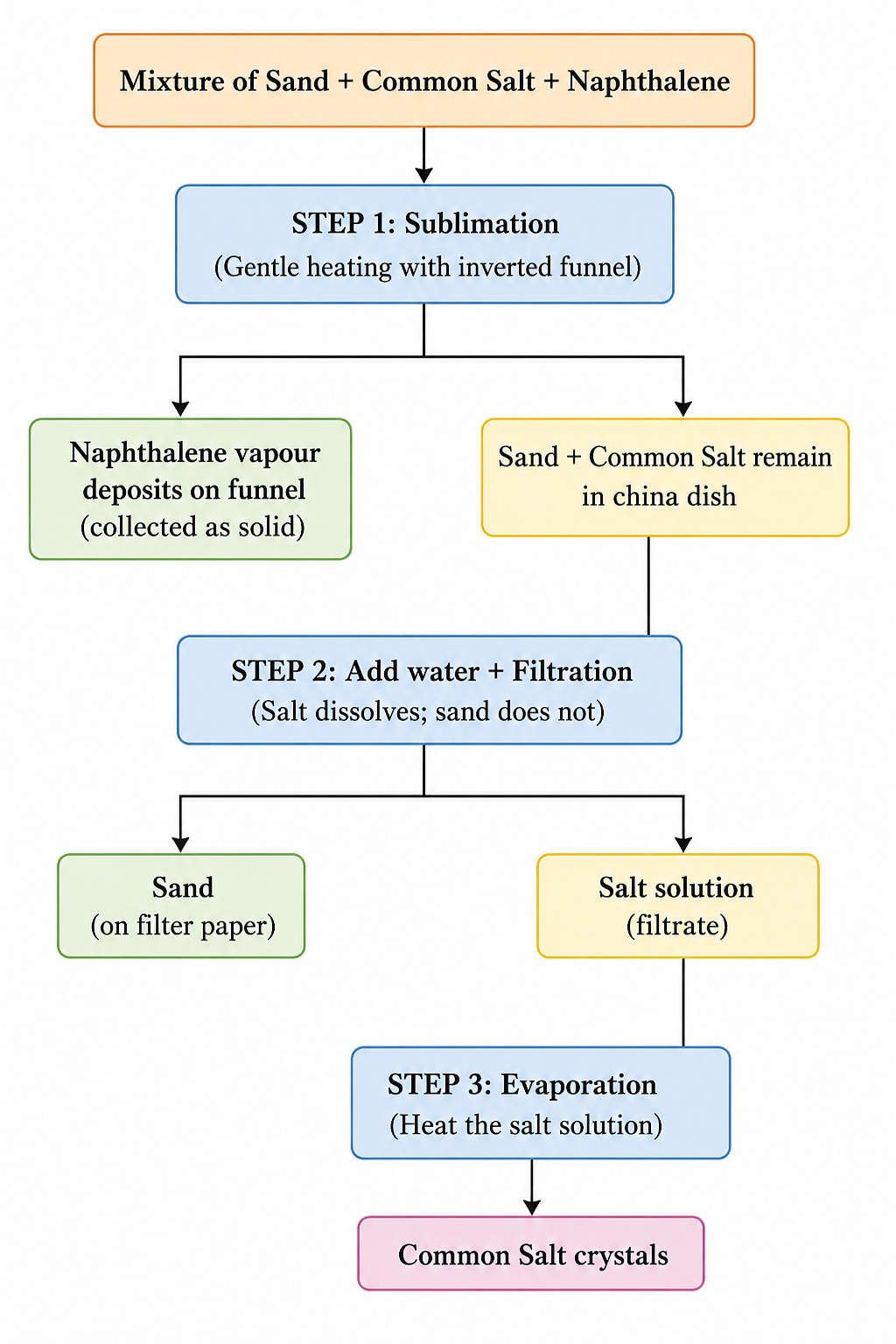
In short: Sublimation → Filtration → Evaporation
- Sublimation separates naphthalene (it sublimes on heating).
- Filtration (after dissolving in water) separates sand from the salt solution.
- Evaporation separates salt from water to give common salt.
Question 12
Why is distillation an effective method for separating a mixture of water and acetone?
Answer
Distillation works here because acetone and water have very different boiling points.
- Boiling point of acetone = 56°C
- Boiling point of water = 100°C
- Difference = 44°C — well above the minimum required 25°C.
When the mixture is heated, acetone vaporises first at 56°C. Very little water vapour forms at this temperature. The acetone vapour is passed through a condenser, cooled, and collected as pure liquid acetone.
Water stays in the distillation flask since it requires a much higher temperature (100°C) to boil.
This large difference in boiling points ensures clean and effective separation of the two liquids.
Question 13
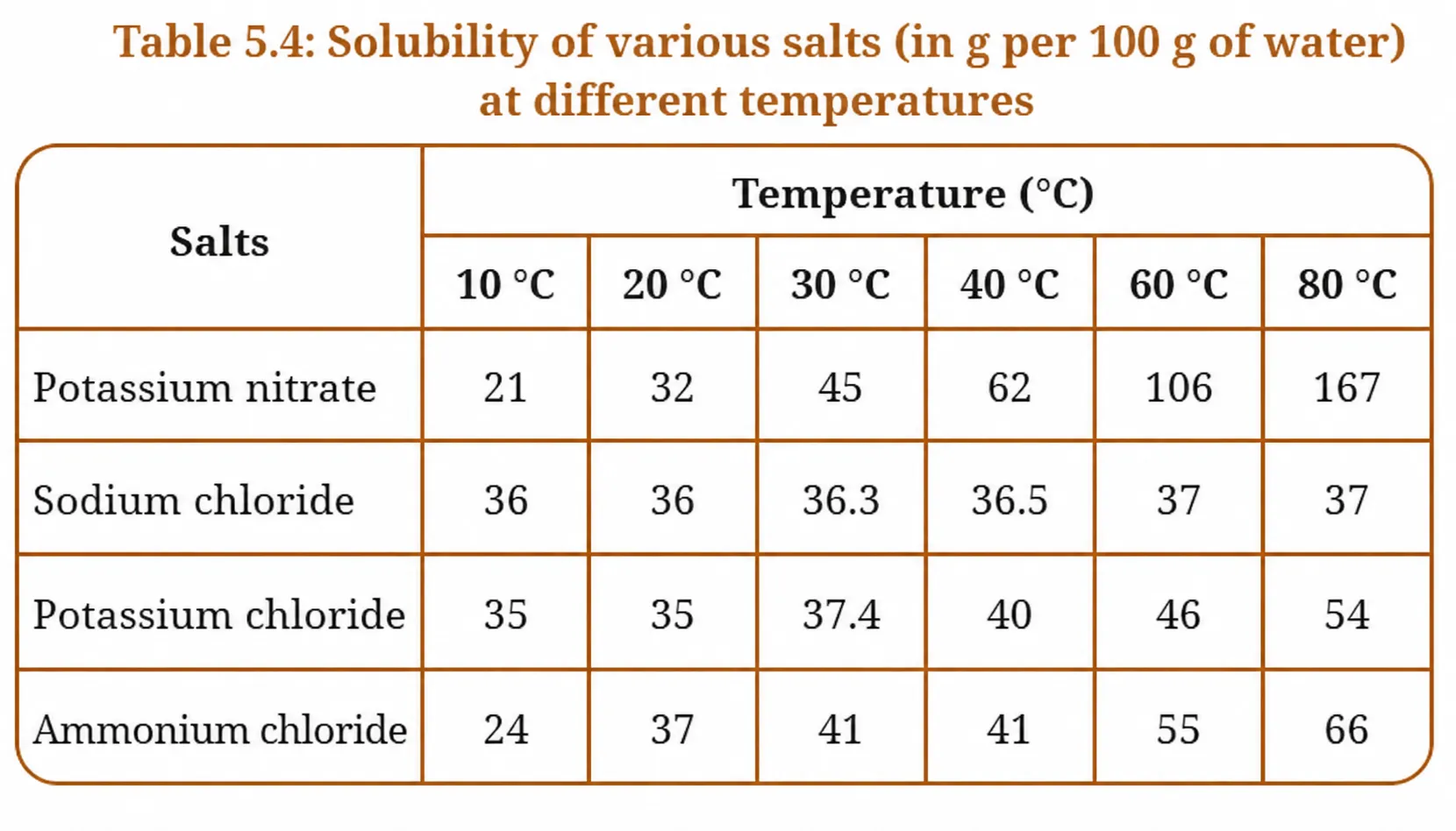
Answer the following questions with the help of the data given in Table 5.4.
Answers
(i) What mass of potassium nitrate would be needed to prepare its saturated solution in 50 g of water at 40°C?
Answer:
From the table, solubility of potassium nitrate at 40°C = 62 g per 100 g of water.
For 50 g of water: mass of KNO₃ needed = (62/100) × 50 = 31 g
(ii) A student makes a saturated solution of potassium chloride in water at 80°C and leaves the solution to cool at room temperature (25°C). What would she observe as the solution cools? Explain.
Answer:
She would observe that crystals of potassium chloride start to form and deposit at the bottom of the beaker as the solution cools.
Reason: At 80°C, the solubility of potassium chloride is 54 g per 100 g of water. At 25°C (approximately 20°C from the table), the solubility drops to only about 35 g per 100 g of water. The solution becomes supersaturated — it holds more dissolved salt than it can at the lower temperature. The excess salt (54 − 35 = ~19 g per 100 g water) therefore precipitates out as crystals.
(iii) What is the effect of a change in temperature on the solubility of salts? Also, compare the changes in the solubility of the four given salts with increasing temperature from 10°C to 80°C.
Answer:
General effect: The solubility of solid solutes in water generally increases with temperature.
Comparison of the four salts:
- Potassium nitrate shows the most dramatic increase — from 21 g at 10°C to 167 g at 80°C. This is nearly an 8-fold increase. It is highly sensitive to temperature.
- Ammonium chloride shows a significant increase — from 24 g at 10°C to 66 g at 80°C.
- Potassium chloride shows a moderate increase — from 35 g at 10°C to 54 g at 80°C.
- Sodium chloride shows almost no change — it remains nearly constant (36 to 37 g) across all temperatures. Temperature barely affects its solubility.
Question 14
Three students, A, B and C, are preparing sugar solutions for an experiment:
● Student A dissolves 20 g of sugar in 80 g of water.
● Student B dissolves 20 g of sugar in 100 g of water.
● Student C dissolves 30 g of sugar in 80 g of water.
(i) Calculate the mass percentage (% m/m) concentration of sugar in
each student’s solution.
(ii) Whose solution is the most concentrated? Explain why.
Answer
(i) Calculations:
Formula: % m/m = (Mass of solute / Total mass of solution) × 100
Student A: Total mass = 20 + 80 = 100 g % m/m = (20/100) × 100 = 20%
Student B: Total mass = 20 + 100 = 120 g % m/m = (20/120) × 100 = 16.67%
Student C: Total mass = 30 + 80 = 110 g % m/m = (30/110) × 100 = 27.27%
(ii) Student C has the most concentrated solution (27.27%).
This is because Student C dissolved the most sugar (30 g) in the least amount of water (80 g). A higher amount of solute in a smaller amount of solvent means a higher concentration. Among the three, this ratio gives the highest mass percentage.
Question 15
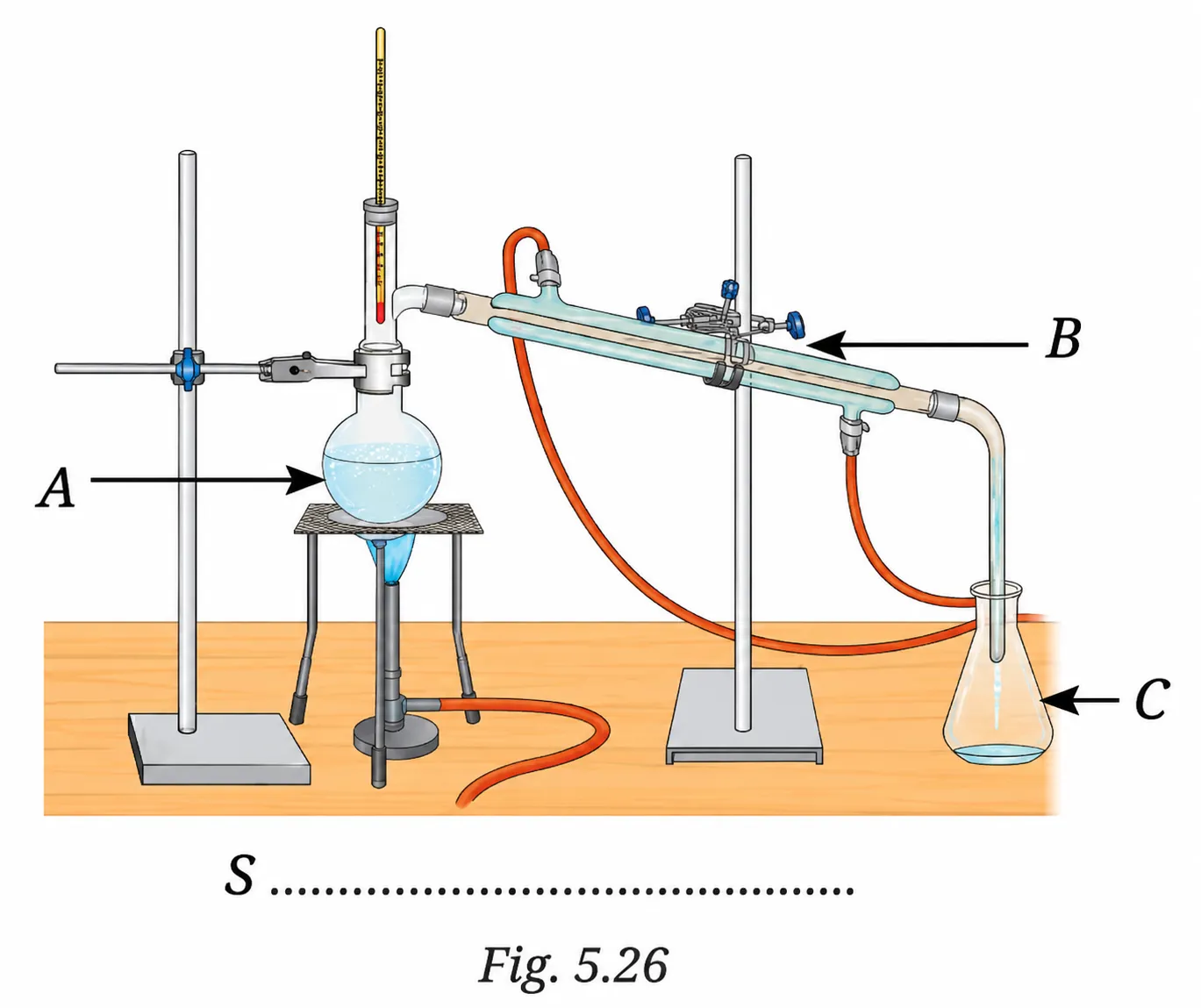
Examine Fig. 5.26.
(i) Identify the separation technique marked as ‘S’.
(ii) Label the apparatus A, B and C.
(iii) Which of the following mixtures can be separated
by the technique identified above? Use the data
given in Table 5.5. Mixtures:
(a) water — acetone (b) water — salt
(c) acetone — alcohol (d) sand — salt
(e) alcohol — chloroform (f) alcohol — benzene

Answer
(i) The separation technique marked as ‘S’ is Distillation.
The figure shows a round-bottomed flask being heated, connected to a condenser that cools the vapour, which then drips into a collecting flask. This is the standard distillation set-up.
(ii) Labels:
- A = Distillation flask (round-bottomed flask containing the mixture, placed on a tripod stand over the burner)
- B = Water condenser (through which cold water flows to cool the vapour)
- C = Conical flask / Collecting flask (collects the distillate — the separated pure liquid)
(iii) Mixtures that can be separated by Distillation:
Distillation requires the two components to be miscible liquids (or a liquid + dissolved solid) with a difference in boiling points of at least ~25°C.
- (a) Water — Acetone ✓ — Difference = 100 − 56 = 44°C → Can be separated
- (b) Water — Salt ✓ — Salt is a solid dissolved in water. Distillation can recover pure water. → Can be separated
- (c) Acetone — Alcohol ✗ — Difference = 78 − 56 = 22°C → Less than 25°C; simple distillation is not effective (fractional distillation would be needed)
- (d) Sand — Salt ✗ — Both are solids; distillation cannot separate solid-solid mixtures. Dissolving in water followed by filtration is better.
- (e) Alcohol — Chloroform ✗ — Difference = 78 − 61 = 17°C → Less than 25°C; simple distillation not effective
- (f) Alcohol — Benzene ✓ — Difference = 80 − 78 = 2°C → Very small difference; simple distillation is not effective
Mixtures that CAN be separated by distillation: (a) and (b)
End of Chapter 5 — Revise, Reflect, Refine Answers.




