Unlock the secrets of atomic structure with our comprehensive Ch 8 Journey Inside the Atom Notes Class 9.
These notes are strictly based on the NCERT Chapter 8, Journey inside the atom, and the CBSE curriculum for class 9 students, covering everything from historical atomic models to modern electron distributions.
Whether you are looking for a quick revision or a deep dive into subatomic particles, this guide simplifies complex concepts to help you score higher in your science exams.
For more Class 9 Notes, Click Here
Rediscovering the Roots of Atomic Theory
Two civilisations, independently, asked: “What is everything made of?”
1. Acharya Kanada (India)
Text: Vaisesika Sutras
➽ Divided matter (dravya) repeatedly ➜ reached the smallest indivisible particle
➽ Called it parmanu
➽ Parmanu is infinitely small and cannot be sensed
➽ Combines into dyads (2 parmanus) & triads (3 parmanus) ➜ forms all matter
2. Leucippus & Democritus (Greece)
● Called indivisible particles, atomos (Greek: indivisible)
A Short Historical Journey Through Atomic Models
Before the late 19th Century
➽ Atoms = the smallest, indivisible units of matter
➽ Then: discovery of radioactivity — certain elements emit energy & particles
➽ This proved that atoms do have smaller particles inside ➜ not indivisible
Discovery of the Electron — J.J. Thomson (1897)
Experiment:
➽ Electric current through a gas at very low pressure
➽ Glass tube with two electrodes + high voltage applied
➽ Observed rays moving: cathode (−) ➜ anode (+) = called cathode rays
Conclusions:
➽ Cathode rays = streams of negatively charged particles (electrons)
➽ Mass of electron << mass of atom
➽ The cathode ray nature was independent of the cathode material or the gas used
∴ Electrons are present in all atoms, in every element
Key fact: Charge of electron = −1.602 × 10⁻¹⁹ C (written as −1 by convention)
Thomson’s model of an atom
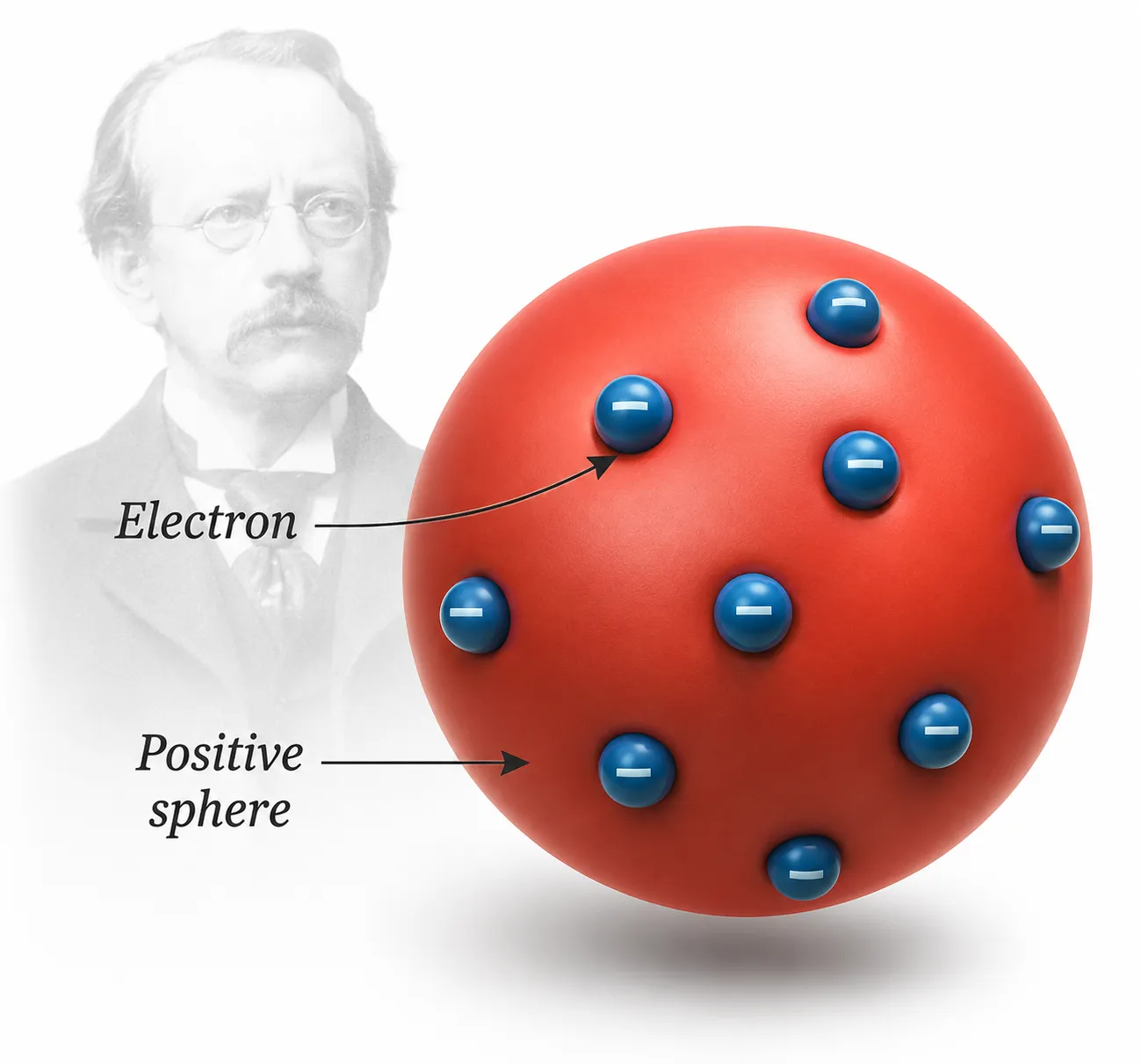
Thomson’s Atomic Model — Plum Pudding Model
The puzzle:
Electrons are negative, but atoms are neutral ➜ where is the positive charge?
Thomson’s solution:
An atom is a sphere of positive charge with electrons distributed throughout it
Analogies used:
🍮 Pudding (positive) with plums (electrons) embedded ➜ Plum pudding model
🍉 Watermelon — red pulp = positive charge, seeds = electrons
Significance:
First genuine attempt to explain how positive and negative charges stay balanced in an atom
Testing Thomson’s model: The gold foil experiment
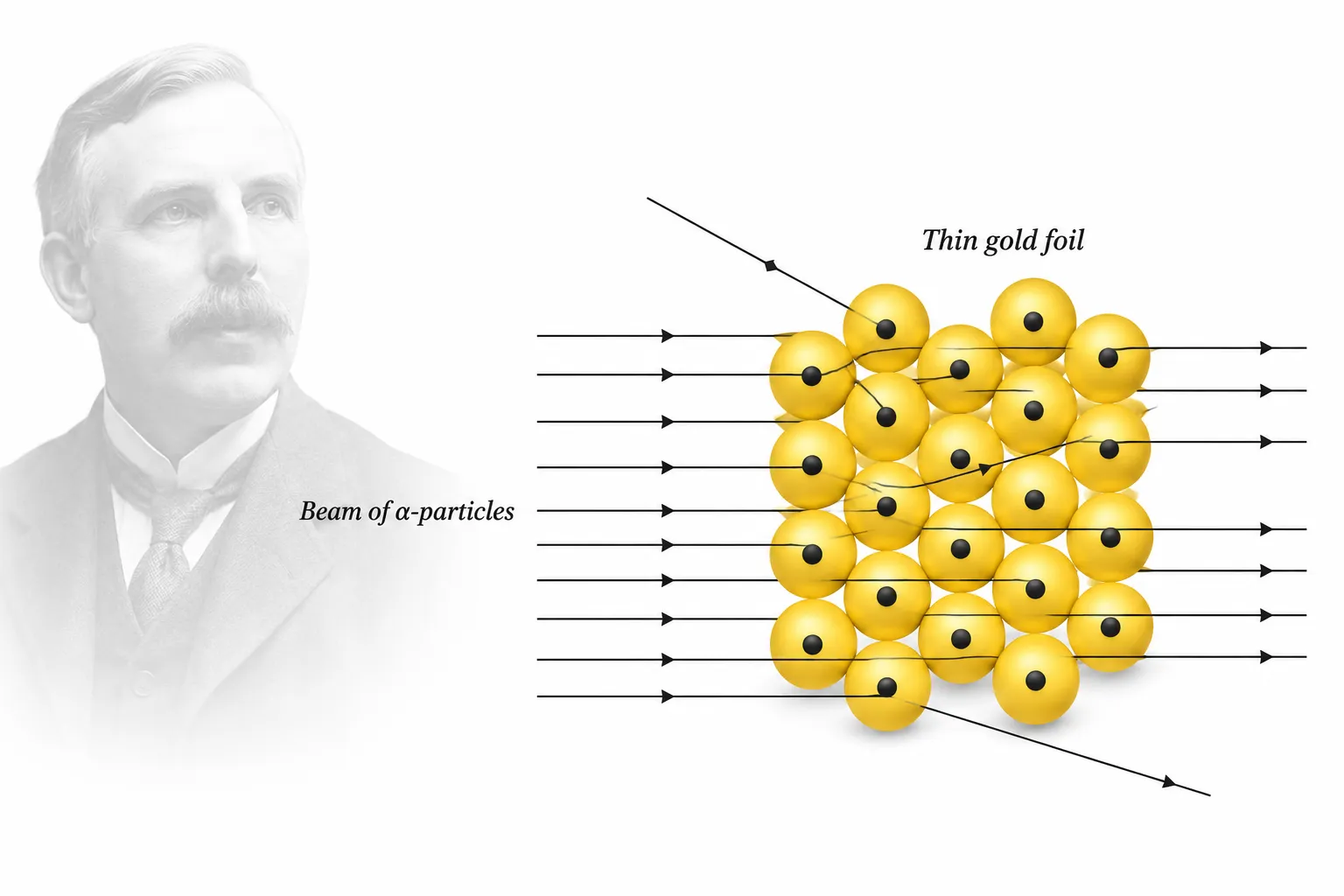
Gold Foil Experiment (1911)
Who: Geiger & Marsden, under Ernest Rutherford.
Purpose: To test Thomson’s atomic model
Setup
➽ A narrow beam of alpha (α) particles fired at a very thin gold foil
➽ α-particle = positively charged, emitted from radioactive elements
(later known: = helium nucleus, 2 protons + 2 neutrons)
Expected vs. Actual Results
| Thomson’s Prediction | Actual Result | |
|---|---|---|
| Most particles | Pass straight through | ✅ Passed undeflected |
| Some particles | Slight deflection | ❌ Sharply deflected |
| Few particles | — | ❌ Bounced straight back |
➽ This deflection from a straight path = scattering
∴ Also called an α-ray scattering experiment
Outcome
- Thomson’s model failed to explain:
- Why are some α-particles deflected at large angles
- Why do most α-particles pass through undeflected
- Results pointed to a fundamentally different atomic structure
A. Rutherford’s model of an atom

suggested by Rutherford| Ch 8 Journey Inside the Atom Notes Class 9
Also called: Planetary Model
Key Proposals
- Most of an atom = space (explains why most α-particles passed through)
- Nucleus = tiny, dense, central region containing:
- All positive charges
- Most of the atom’s mass
- Electrons revolve around the nucleus like planets around the Sun
Size Comparison
| Entity | Diameter |
|---|---|
| Atom | ≈ 10⁻¹⁰ m |
| Nucleus | ≈ 10⁻¹⁵ m |
The nucleus is 10⁵ (one lakh) times smaller than the atom
🏏 Analogy: If atom = cricket ground (100 m), nucleus = a pepper grain at the centre
vs. Thomson’s Model
➽ Better — successfully explained gold foil experiment results
➽ Limitation — could not explain the stability of the atom
B. Limitations of Rutherford’s model
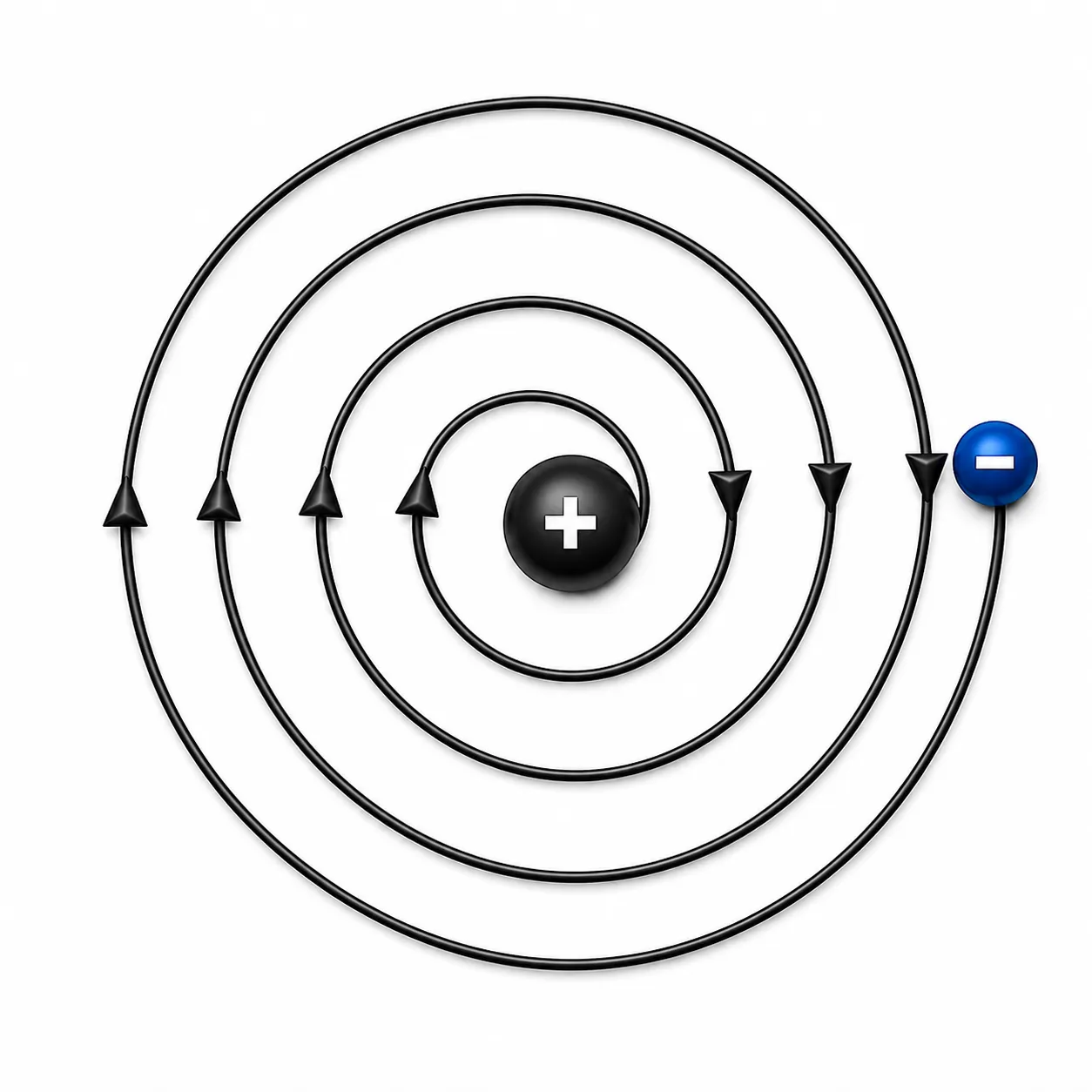
charged particle on losing energy| Ch 8 Journey Inside the Atom Notes Class 9
The Stability Problem
The logic:
➽ Electron moves in a circular path ➜ constantly changing direction ➜ accelerating
➽ An accelerating charged particle loses energy
➽ Losing energy ➜ electron spirals inward ➜ falls into nucleus
∴ The atom should collapse
The reality:
➽ Atoms are stable — matter around us stays intact
➽ Rutherford’s model couldn’t explain this contradiction
∴ A new explanation was needed for how electrons stay in motion without collapsing into the nucleus
C. Discovery of the proton
Facts
- Nucleus carries a positive charge
➜ due to particles called protons - Protons are much heavier than electrons
- Charge of proton = equal and opposite to electron (+1)
Electrical Neutrality of Atoms
For an atom to be neutral:
no. of protons = no. of electrons
Examples:
| Atom | Protons | Electrons |
|---|---|---|
| Helium | 2 | 2 |
| Sodium | 11 | 11 |
Total +ve charge = Total −ve charge ➜ atom is electrically neutral
This holds for all atoms
Bohr’s model of the atom
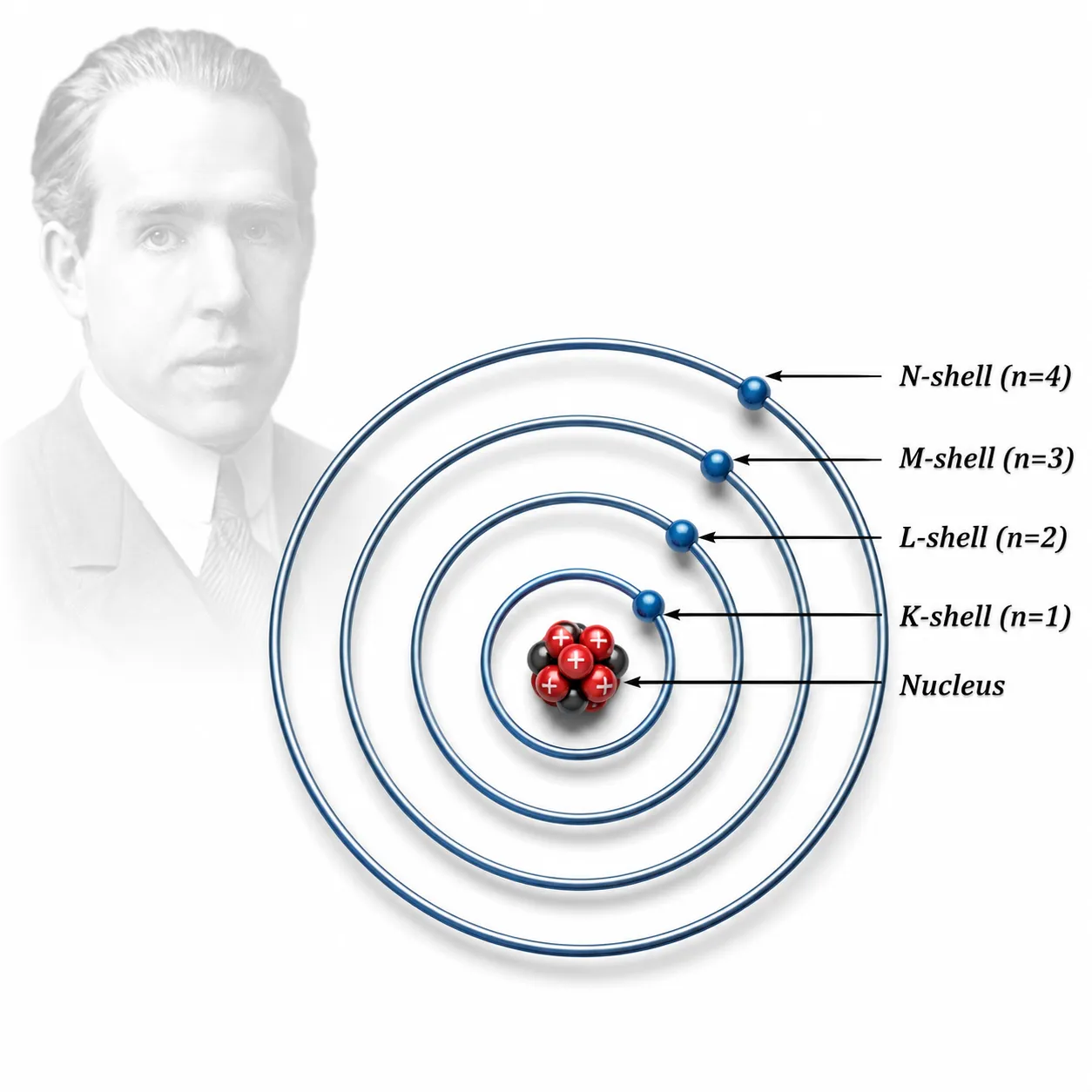
atom| Ch 8 Journey Inside the Atom Notes Class 9
Purpose: Explain why atoms are stable
Postulates
➽ Electrons move in fixed circular paths around the nucleus
➽ These paths = stationary states/orbits/shells/energy levels
➽ Each shell has a definite, constant energy
➽ Electrons do not lose energy while in a fixed shell
Shells & Energy Levels
➽ Shells: K, L, M, N… or n = 1, 2, 3, 4…
➽ K-shell (n=1) = closest to nucleus = lowest energy
➽ Energy increases as we move away from the nucleus
➽ Each shell holds only a certain number of electrons
➽ Electrons can only exist in shells, not between them
Electron Transitions
➽ Electrons move between shells by absorbing or releasing energy
➽ Energy absorbed/released = exact difference between the two energy levels
How It Explains the Stability of Atoms
➽ Rutherford’s problem: the moving electron should lose energy & collapse
➽ Bohr’s fix: introduced stationary states as a postulate
➽ In a stationary state ➜ energy remains constant even while in motion
∴ No energy loss ➜ no collapse ➜ atom stays stable
Bohr’s model explained many experimental observations and was
a major step in understanding atomic structure.
What Components Contribute to the Mass of an Atom?
The Puzzle
➽ Helium has 2 protons, yet its mass = 4× that of hydrogen (not 2×)
∴ Something else in the nucleus was adding mass without charge
Discovery of the Neutron
By James Chadwick (1932)
➽ Chadwick (student of Rutherford) discovered a new subatomic particle
➽ Neutron (n) = mass ≈ proton, but no charge
➽ Found in the nucleus of all atoms except hydrogen
∴ Atomic mass comes mainly from protons + neutrons in the nucleus
Subatomic particles
| Particle | Symbol | Relative Charge |
|---|---|---|
| Electron | e⁻ | −1 |
| Proton | p⁺ | +1 |
| Neutron | n⁰ | 0 |
Symbols of Elements
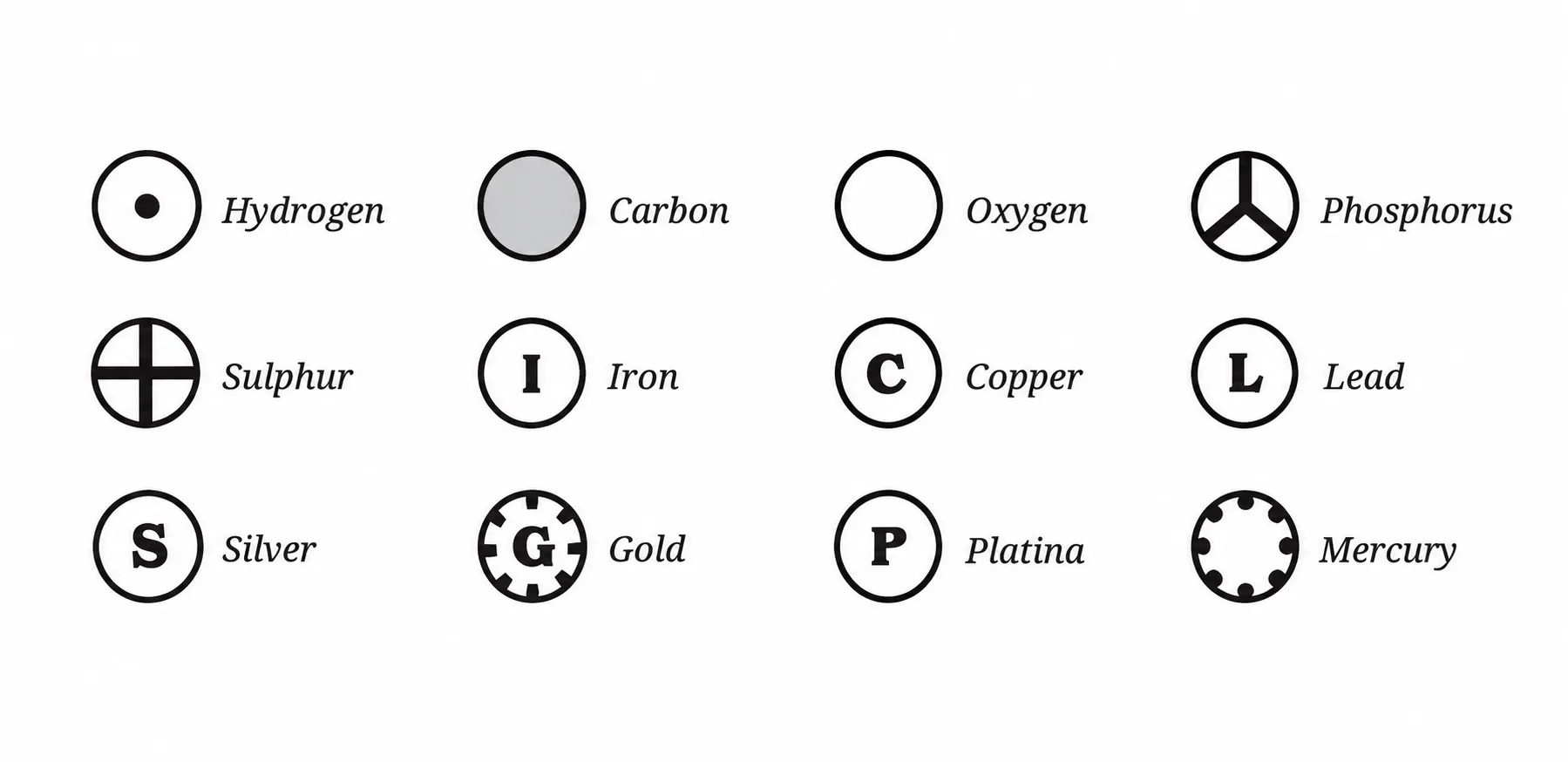
1869: 69 elements known ➜ Today: 118 elements
Why Symbols?
Internationally recognised ➜ scientists worldwide can communicate regardless of language
Common Elements & Their Symbols
| Element | Symbol | Element | Symbol |
|---|---|---|---|
| Aluminium | Al | Magnesium | Mg |
| Argon | Ar | Neon | Ne |
| Barium | Ba | Nitrogen | N |
| Boron | B | Oxygen | O |
| Bromine | Br | Silicon | Si |
| Calcium | Ca | Sulfur | S |
| Carbon | C | Uranium | U |
| Chlorine | Cl | Zinc | Zn |
| Cobalt | Co | Fluorine | F |
| Hydrogen | H | Iodine | I |
Symbols from Other Languages
Fe ➜ Iron (Latin: ferrum)
Hg ➜ Mercury (Greek: hydrargyros)
W ➜ Tungsten (German: Wolfram)
Au ➜ Gold (Latin: aurum)
K ➜ Potassium (Latin: kalium)
Na ➜ Sodium (Latin: natrium)
Ag ➜ Silver (Latin: argentum)
Pb ➜ Lead (Latin: plumbum)
Cu ➜ Copper (Latin: cuprum)
Atomic Number
Definition:
The number of protons in the nucleus of an atom is called its atomic number, designated by the symbol Z
➽ Z determines the identity of an element and its chemical behaviour
➽ Since atoms are neutral: no. of protons = no. of electrons
➽ Each element has a unique Z ➜ no two elements share the same atomic number
Examples
| Element | Protons | Electrons | Z |
|---|---|---|---|
| Hydrogen | 1 | 1 | 1 |
| Helium | 2 | 2 | 2 |
| Lithium | 3 | 3 | 3 |
Key point:
Atomic number uniquely identifies an element — elements with different atomic numbers are always distinct from each other.
Mass Number
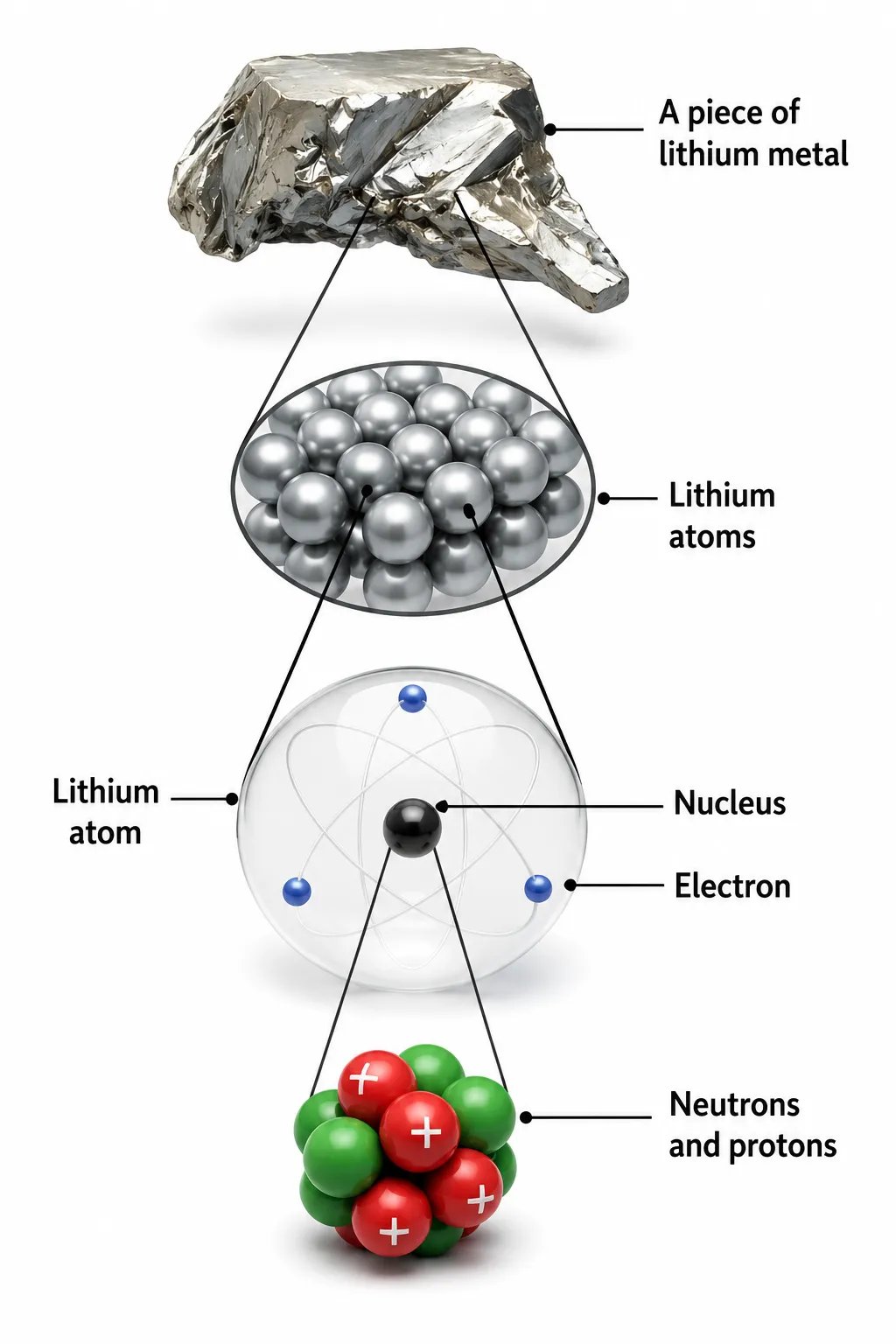
in the nucleus| Ch 8 Journey Inside the Atom Notes Class 9
Definition
The total number of protons and neutrons in the nucleus is called the mass number, denoted by A
➽ Protons + Neutrons in nucleus = Nucleons
➽ A = no. of protons + no. of neutrons
➽ Electron mass is negligible ➜ ignored in calculations
Examples
| Element | Protons (p⁺) | Neutrons (n⁰) | Mass No. (A) |
|---|---|---|---|
| Hydrogen | 1 | 0 | 1 |
| Helium | 2 | 2 | 4 |
| Lithium | 3 | 4 | 7 |
Standard Atomic Notation
Mass number on top, Atomic number at bottom, Symbol in middle
Example — Carbon:
- Symbol = C, Z = 6, A = 12
- Written as: ¹²₆C
How Are Electrons Distributed in Different Energy Levels?
Bohr-Bury Rules
1. Max electrons per shell = 2n²
| Shell | n | Max electrons (2n²) |
|---|---|---|
| K | 1 | 2 |
| L | 2 | 8 |
| M | 3 | 18 |
2. Outermost shell can hold max 8 electrons (except K-shell ➜ max 2)
3. Filling order — stepwise, from nucleus outward: K ➜ L ➜ M ➜ N…
- Next shell fills only after the previous one is complete
Examples
Hydrogen (Z = 1)
- 1 electron ➜ goes into K-shell
- Distribution: K = 1
Helium (Z = 2)
- 2 electrons ➜ both fit in K-shell (max = 2)
- Distribution: K = 2
Key point:
Electrons always fill the innermost available shell first before moving to the next.
Building up atoms
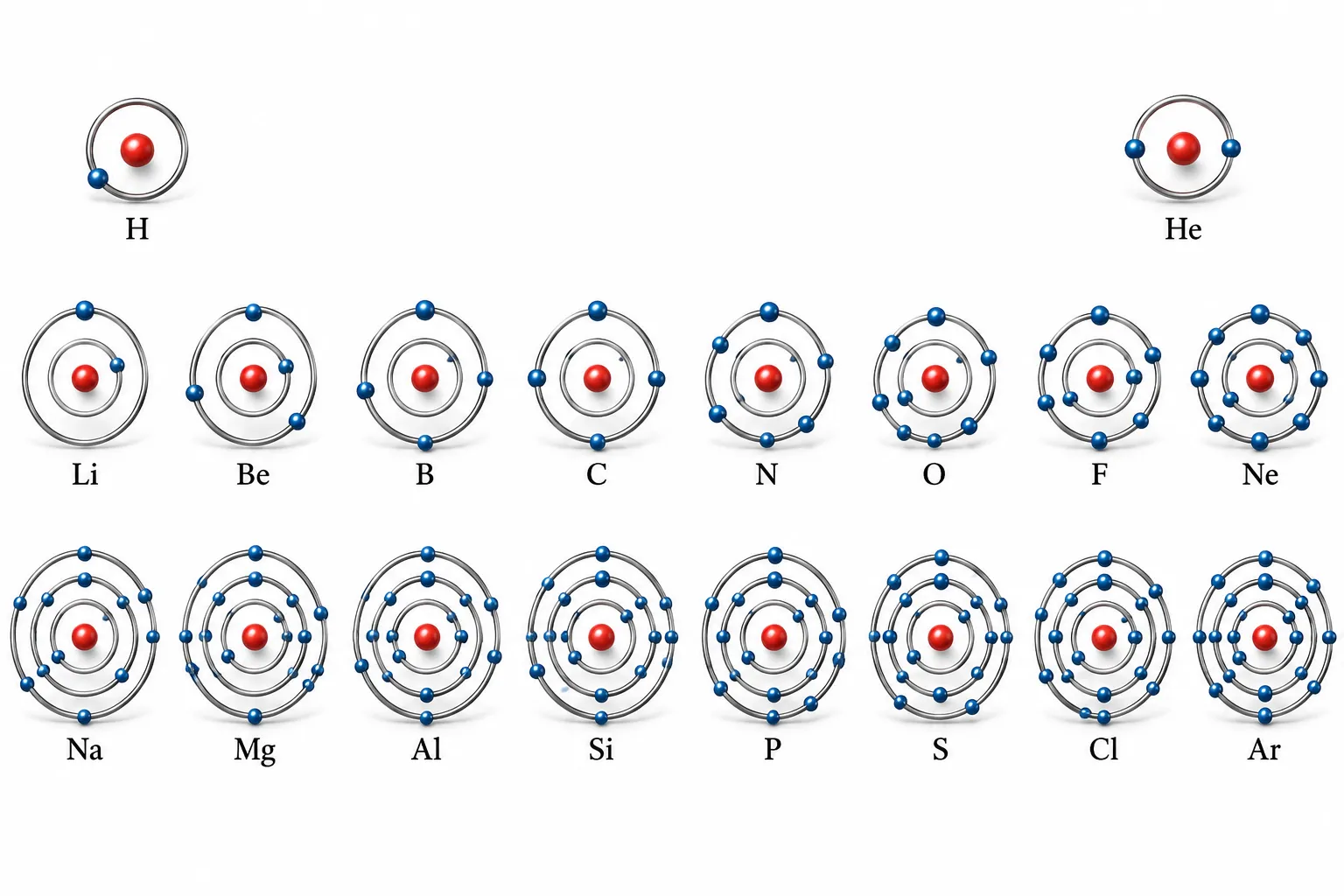
are filled in the K, L and M shells| Ch 8 Journey Inside the Atom Notes Class 9
Electronic Configuration
The distribution of electrons among various shells of an atom is called its electronic configuration
➽ Built by adding one electron for each increase in atomic number
➽ Electrons fill shells in order: K ➜ L ➜ M ➜ N…
➽ Follow Bohr-Bury rules for max electrons per shell
Maximum Electrons Per Shell: The formula calculates the capacity, where “n” is the shell number (K, L, M, N…):
Symbols, atomic numbers, number of protons, number of neutrons, number of
electrons, and the electronic distribution of atoms of the first eighteen elements
Click on the elements to reveal the details
Combining Capacity of an Atom: Valency
Definitions
➽ Valence shell = outermost shell containing electrons
➽ Valence electrons = electrons present in the valence shell
➽ Valency = number of electrons lost, gained, or shared to complete the octet
Octet Rule
➽ Octet = 8 electrons in valence shell ➜ stable & unreactive
➽ Exception: Helium ➜ stable with just 2 electrons
➽ Incomplete valence shell ➜ atom is reactive
➽ Atoms lose, gain, or share electrons to complete their octet
How Valency is Determined
| Valence electrons | Tendency | Valency |
|---|---|---|
| Less than 4 | Loses electrons | = no. of valence e⁻ |
| More than 4 | Gains electrons | = 8 − valence e⁻ |
| Exactly 4 | Shares electrons | 4 |
Examples
| Element | Config | Valence e⁻ | Action | Valency |
|---|---|---|---|---|
| Sodium | 2,8,1 | 1 | Loses 1 | 1 |
| Oxygen | 2,6 | 6 | Gains 2 | 2 |
| Carbon | 2,4 | 4 | Shares 4 | 4 |
Combining Capacity
➽ Measured against H or Cl (both have a combining capacity of 1)
➽ H₂O ➜ O combines with 2H ➜ combining capacity of O = 2
➽ NH₃ ➜ N combines with 3H ➜ valency of N = 3
➽ MgCl₂ ➜ Mg combines with 2Cl ➜ valency of Mg = 2
Note: Atoms with a complete octet already (noble gases) neither lose nor gain electrons ➜ valency = 0
A Deeper Look into Atomic Structure
Isotopes
Definition
Atoms of the same element with the same atomic number (Z) but different mass numbers (A) due to different numbers of neutrons are called isotopes
Same protons, different neutrons ➜ different mass
Called ‘twin atoms’
Examples
Hydrogen — 3 isotopes
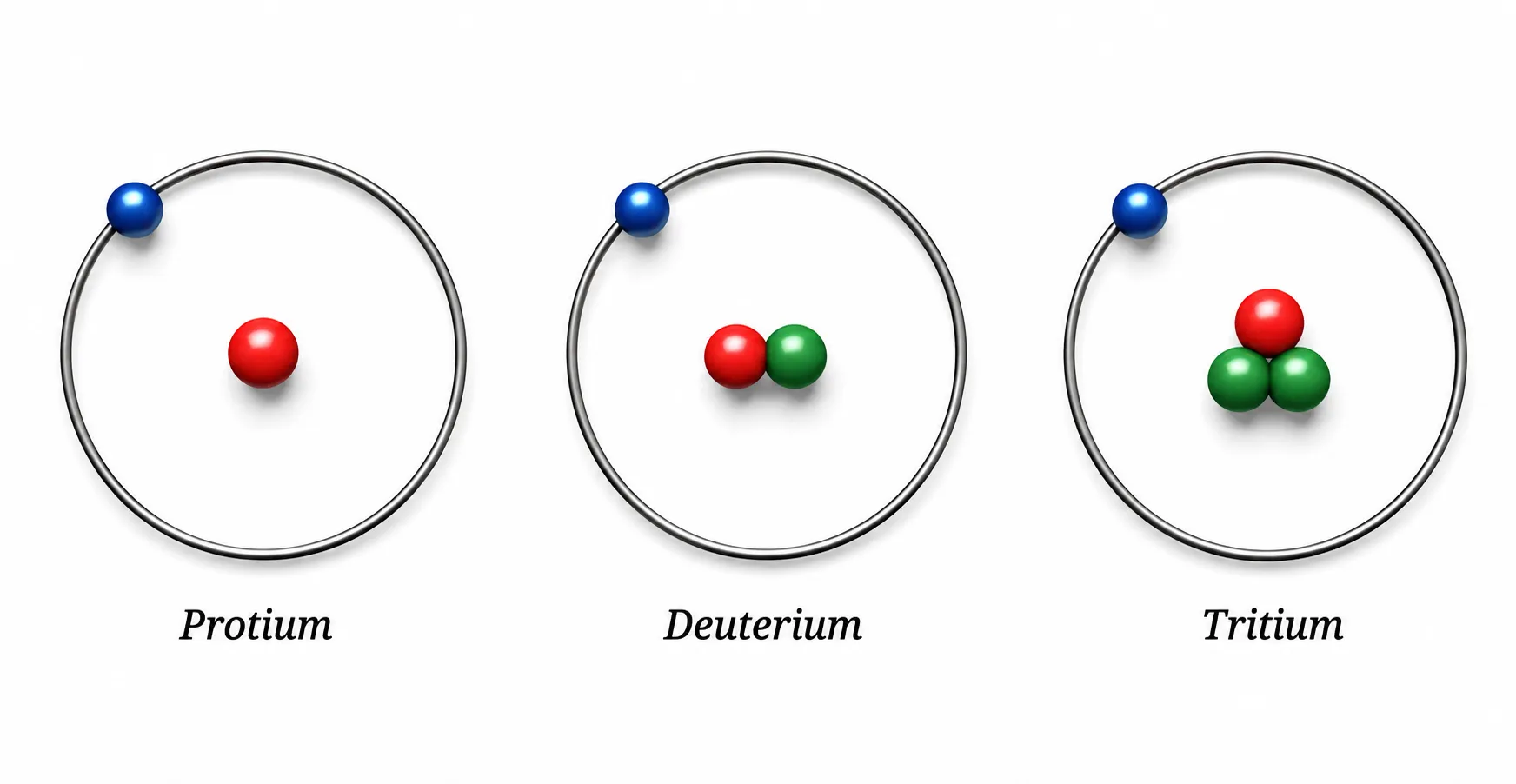
| Isotope | Protons | Neutrons | Electrons |
|---|---|---|---|
| Protium ¹₁H (~99.98%) | 1 | 0 | 1 |
| Deuterium ²₁H (~0.015%) | 1 | 1 | 1 |
| Tritium ³₁H (traces) | 1 | 2 | 1 |
Carbon — 3 isotopes
- All have 6 protons & 6 electrons,
differ only in neutrons - ¹²₆C (most abundant), ¹³₆C, ¹⁴₆C
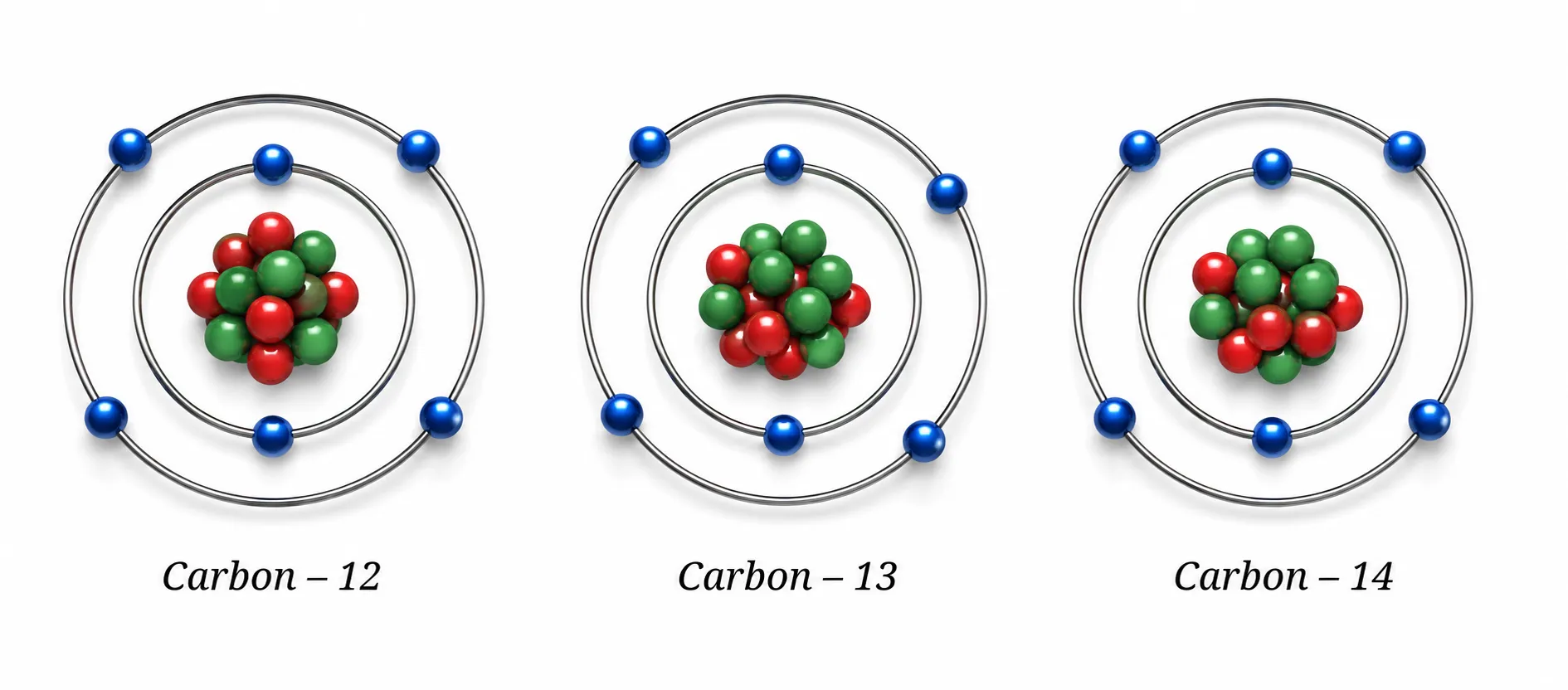
Properties of Isotopes
➽ Chemical properties ➜ same (same electrons, same electronic config, same valence electrons)
➽ Physical properties ➜ different (e.g. boiling point, melting point)
Uses of Isotopes
| Isotope | Use |
|---|---|
| ²³⁵₉₂U (Uranium) | Fuel in nuclear reactors |
| ⁶⁰₂₇Co (Cobalt) | Radiation treatment for cancer |
| ¹³¹₅₃I (Iodine) | Treats goitre & thyroid cancer |
| ¹⁴₆C (Carbon) | Dating ancient fossils & artefacts |
A. Average atomic mass
The Problem
Chlorine has two isotopes: ³⁵Cl and ³⁷Cl ➜ which mass do we use?
Simple Average (Inaccurate)
- Assumes equal abundance of all isotopes
- For Cl: (35 + 37) ÷ 2 = 36 u
- ❌ Doesn’t reflect nature — isotopes don’t occur equally
Weighted Average (Accurate)
- Accounts for the natural
abundance of each isotope - Formula:
Σ (mass of isotope × % abundance) ÷ 100
For Chlorine:
- 35Cl ➜ ~75% abundant
- 37Cl ➜ ~25% abundant
What Does 35.5 u Mean?
➽ No single Cl atom has a mass of 35.5 u
➽ It means: in 10 lakh Cl atoms ➜ 7.5 lakh are ³⁵Cl + 2.5 lakh are ³⁷Cl
➽ 35.5 u = the statistical average across all naturally occurring Cl atoms
Simple vs Weighted Average
| Simple Average | Weighted Average | |
|---|---|---|
| Considers abundance | ❌ No | ✅ Yes |
| Accurate | ❌ No | ✅ Yes |
| Value for Cl | 36 u | 35.5 u |
Isobars
Definition
Atoms of different elements with the same mass number (A) but different atomic numbers (Z) are called isobars
Example:
| Element | Atomic No. (Z) | Mass No. (A) |
|---|---|---|
| Argon (Ar) | 18 | 40 |
| Potassium (K) | 19 | 40 |
| Calcium (Ca) | 20 | 40 |
Different elements, different protons ➜ but same total nucleons
Isotopes vs Isobars
| Isotopes | Isobars | |
|---|---|---|
| Atomic No. (Z) | Same | Different |
| Mass No. (A) | Different | Same |
| Element | Same | Different |
Beyond Bohr — Where the Story Goes
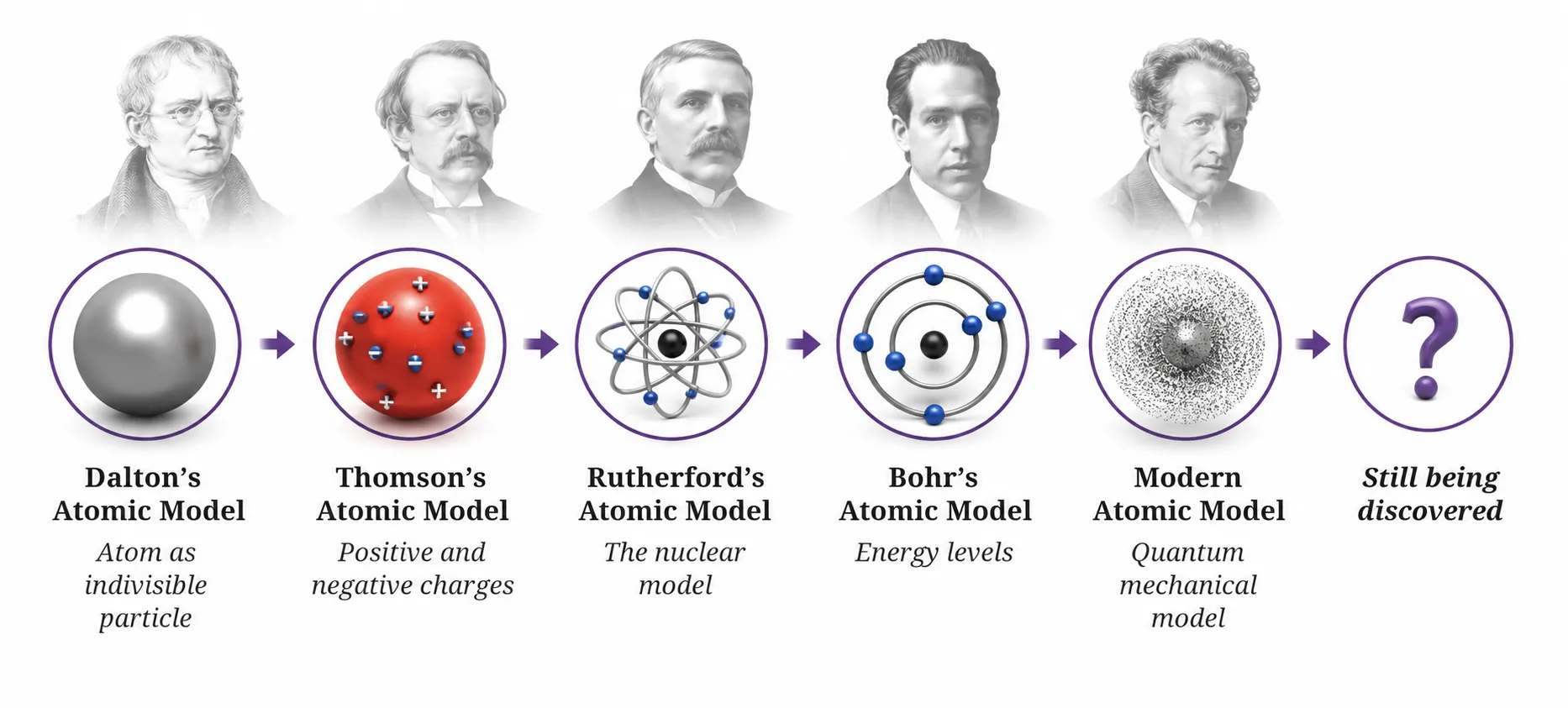
➽ Bohr’s model was a major step, but not entirely correct
➽ Electrons don’t follow fixed, well-defined paths
➽ Modern view: electrons exist as electron clouds around the nucleus
vWe can only predict regions of probability, not exact positions
The exploration of atomic structure is still ongoing — exciting discoveries still lie ahead!
Can You Discover ?



