Chapter 9, Atomic Foundations of Matter, grade 9, is the foundational chapter to understand chemistry.
You must master the basics to understand the formation of molecules and compounds. The chapter has many activities and texts, so students get lost in it and tend to overlook or misread the important points and concepts.
“Ch 9 Atomic Foundations of Matter Notes Class 9” is the solution to that very problem you face while reading the chapter.
In “Ch 9 Atomic Foundations of Matter Notes Class 9”, we have included all the important concepts and points relevant to your examinations.
Revise again and again to score well in your tests and exams.
Happy reading!
Mass, Physical, and Chemical Changes
Physical Changes
NO new substances are formed
➽ Mass before = Mass after
➽ Example: mass of water + salt = mass of solution formed
➽ No change in mass during physical changes
Chemical Changes
New substances are formed
➽ Reaction: Vinegar + Baking soda ➜ Carbon dioxide + Other substances
➽ Total mass before reaction = Total mass after reaction
Conclusion:
Mass is conserved in both physical and chemical changes. The apparent loss in open systems is due to escaped gases, not actual mass loss.
Law of Conservation of Mass
“Matter can neither be created nor destroyed in a chemical reaction.”
Simply:
Total mass of reactants = Total mass of products
Proposed by:
Antoine Lavoisier, 1789
Reaction Examples
- Na₂SO₄ + BaCl₂ ➜ BaSO₄ (white precipitate) + NaCl
- CaCO₃ + HCl ➜ CO₂ + H₂O + CaCl₂
Verify Conservation of Mass
| Side | Substances | Mass |
|---|---|---|
| Reactants | CaCO₃ + HCl | 4.0 + 2.92 = 6.92 g |
| Products | CO₂ + H₂O + CaCl₂ | 1.76 + 0.72 + 4.44 = 6.92 g |
✓ Mass conserved
Q. 12 g of carbon combines with 32 g of oxygen to form 44 g of carbon dioxide as per the given equation.
Carbon + Oxygen ➜ Carbon dioxide
If 2.4 g of carbon reacts completely with oxygen, how much carbon dioxide will be produced?
Solution:
Find the mass of the product using the ratio
12 g C + 32 g O₂ ➜ 44 g CO₂
For 2.4 g C:
(44 ÷ 12) × 2.4 = 8.8 g CO₂ Ans
Law of Constant Proportions
“In any compound, elements always combine in a fixed ratio by mass, regardless of its source.“
Proposed by:
Joseph Proust
Also called:
Law of Definite Proportions / Proust’s Law
Example — Water (H₂O):
H : O = 1 : 8 (by mass), always
9 g of water ➜ 1 g H + 8 g O, every time
Connection to Dalton’s Atomic Theory
These two laws together (Conservation of Mass + Constant Proportions)
formed the basis of Dalton’s Atomic Theory.
Dalton explained both laws by stating:
➽ Atoms are indivisible during a chemical reaction
➽ Dalton explained both laws by stating:
↪ Atoms are indivisible during a chemical reaction
↪ They simply rearrange to form new substances
This explains why mass is conserved and why compounds always have fixed elemental ratios.
Dalton’s Atomic Theory
Proposed by:
John Dalton
What is Postulate:
“A fundamental assumption accepted as truth without formal proof, from which further ideas are formed or developed.“
Postulates Of Dalton’s Atomic Theory
➊All matter is made of very tiny particles called atoms, which participate in chemical reactions
➋Atoms are indivisible ➜ cannot be created or destroyed in a reaction
➌Atoms of the same element ➜ identical in mass & chemical properties
➍Atoms of different elements ➜ different masses & chemical properties
➎Atoms combine in simple whole-number ratios to form compounds
➏The relative number and kinds of atoms are constant in a given compound
Examples
H + O ➜ Water (atoms rearrange, not destroyed)
Mg + O ➜ MgO (magnesium atoms combine with oxygen atoms)
Significance Of Dalton’s Atomic Theory
➽ Explains why mass is conserved (atoms only rearrange)
➽ Explains fixed ratios in compounds (atoms combine in whole numbers)
➽ Forms the basis for the modern understanding of atoms and their behaviour
➽ Later led scientists to discover how atoms combine to form molecules
How Atoms Combine?
Molecule
“An electrically neutral entity consisting of more than one atom, capable of independent existence, showing all properties of that substance.“
Properties of a molecule:
➽ Same element atoms ➜ molecule of element (e.g., H + H ➜ H₂)
➽ Different element atoms ➜ molecule of compound (e.g., H + Cl ➜ HCl)
➽ Some elements like Helium exist only as atoms ➜ already stable, don’t combine
Why Atoms Combine ➜ The Octet Rule:
Atoms are stable when they have:
➽ 8 electrons (Octet) in the outermost (valence) shell, OR
➽ 2 electrons if the K-shell is the outermost.
If the valence shell is incomplete ➜ atoms combine to become stable
How They Combine
1. Sharing of electrons
● Atoms share some or all valence electrons with another atom
2. Transfer of electrons
● An atom gives away one or more electrons,
OR
● An atom accepts one or more electrons from another atom
Chemical Bond
➽ When atoms combine, the total energy of the system decreases → more stable arrangement
➽ The force that holds atoms together = Chemical Bond
Main idea:
Atoms combine to achieve stability by completing their valence shell
➜ either by sharing or transferring electrons.
Bonding by sharing of electrons — Covalent Bond
“The interaction between atoms through a shared pair of electrons is called a covalent bond.“
A. Molecules of elements
Types of Covalent Bonds
| Bond Type | Shared Pairs | Example | Representation |
|---|---|---|---|
| Single bond | 1 pair | H₂, Cl₂ | H—H, Cl—Cl |
| Double bond | 2 pairs | O₂ | O=O |
How Each Molecule Forms
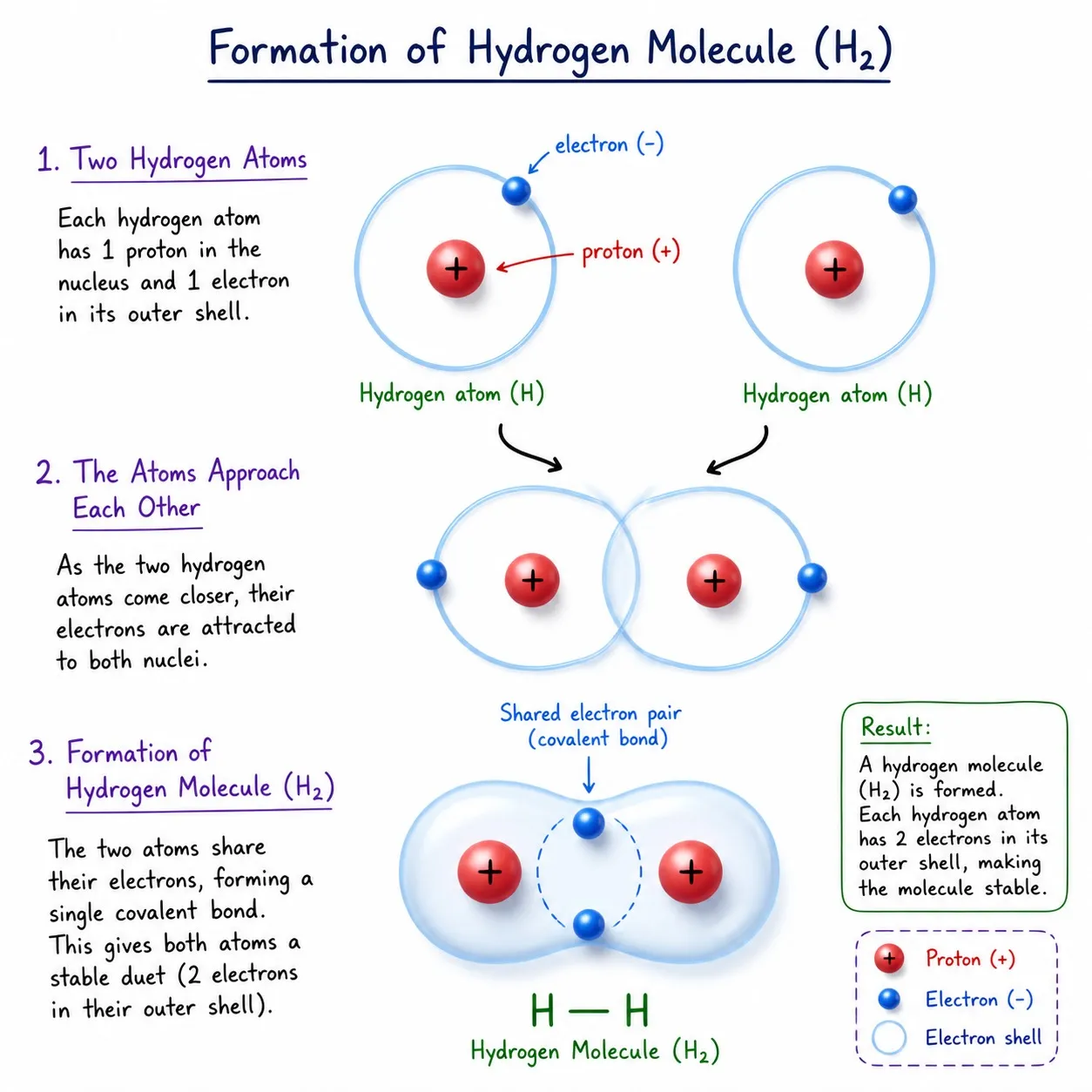
Hydrogen (H₂)
➽ H has 1 electron in K-shell; needs 1 more (K-shell max = 2)
➽ Two H atoms share 1 electron each ➜ H—H
➽ Shared pair attracts both nuclei ➜ stable molecule
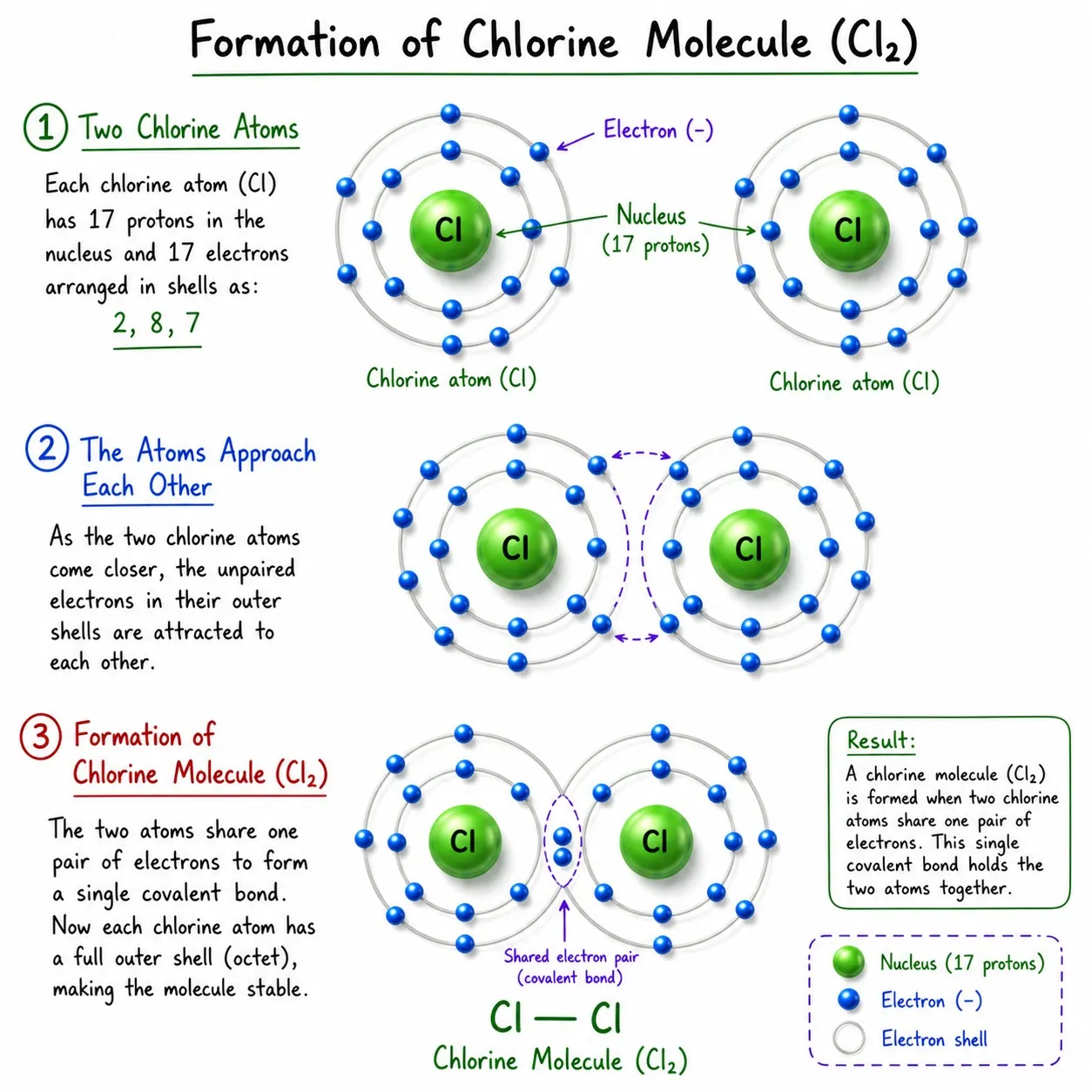
Chlorine (Cl₂)
➽ Cl has 7 valence electrons; needs 1 more
➽ Two Cl atoms share 1 electron each ➜ Cl—Cl
➽ Single covalent bond
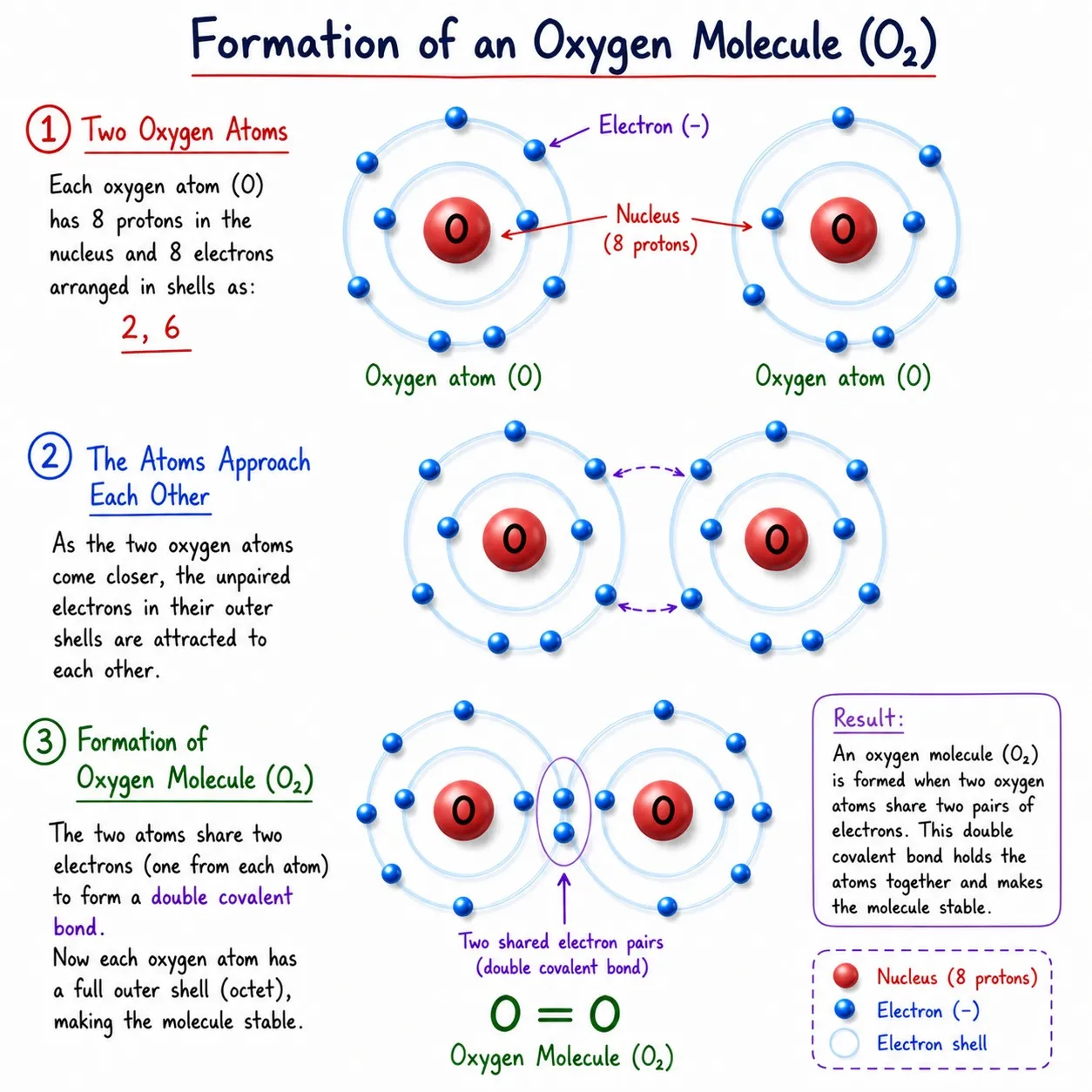
Oxygen (O₂)
➽ O has 6 valence electrons; needs 2 more
➽ Two O atoms share 2 electrons each ➜ O=O
➽ Double bond (2 shared pairs)
Main idea
- Atoms share electrons to complete their valence shell
- Shared pair attracts both nuclei ➜ holds molecule together
- More shared pairs = stronger/different bond type
B. Molecules of compounds
When atoms of two different elements share electrons, ➜ forms a covalent compound
How Each Molecule Forms
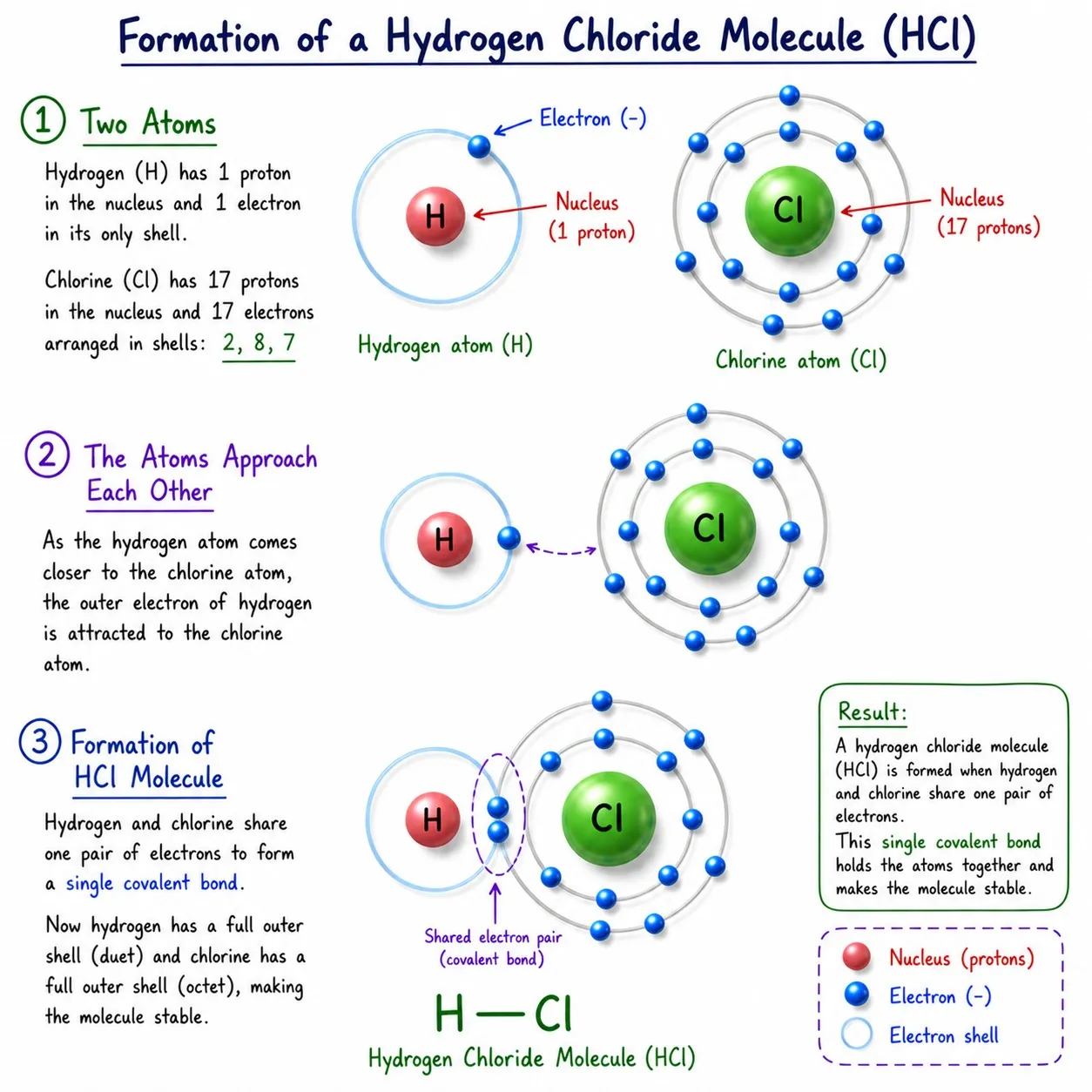
Hydrogen Chloride (HCl)
➽ H ➜ 1 valence electron, needs 1 more
➽ Cl ➜ 7 valence electrons, needs 1 more
➽ Both need 1 electron ➜ share 1 electron each
➽ Single covalent bond ➜ H—Cl
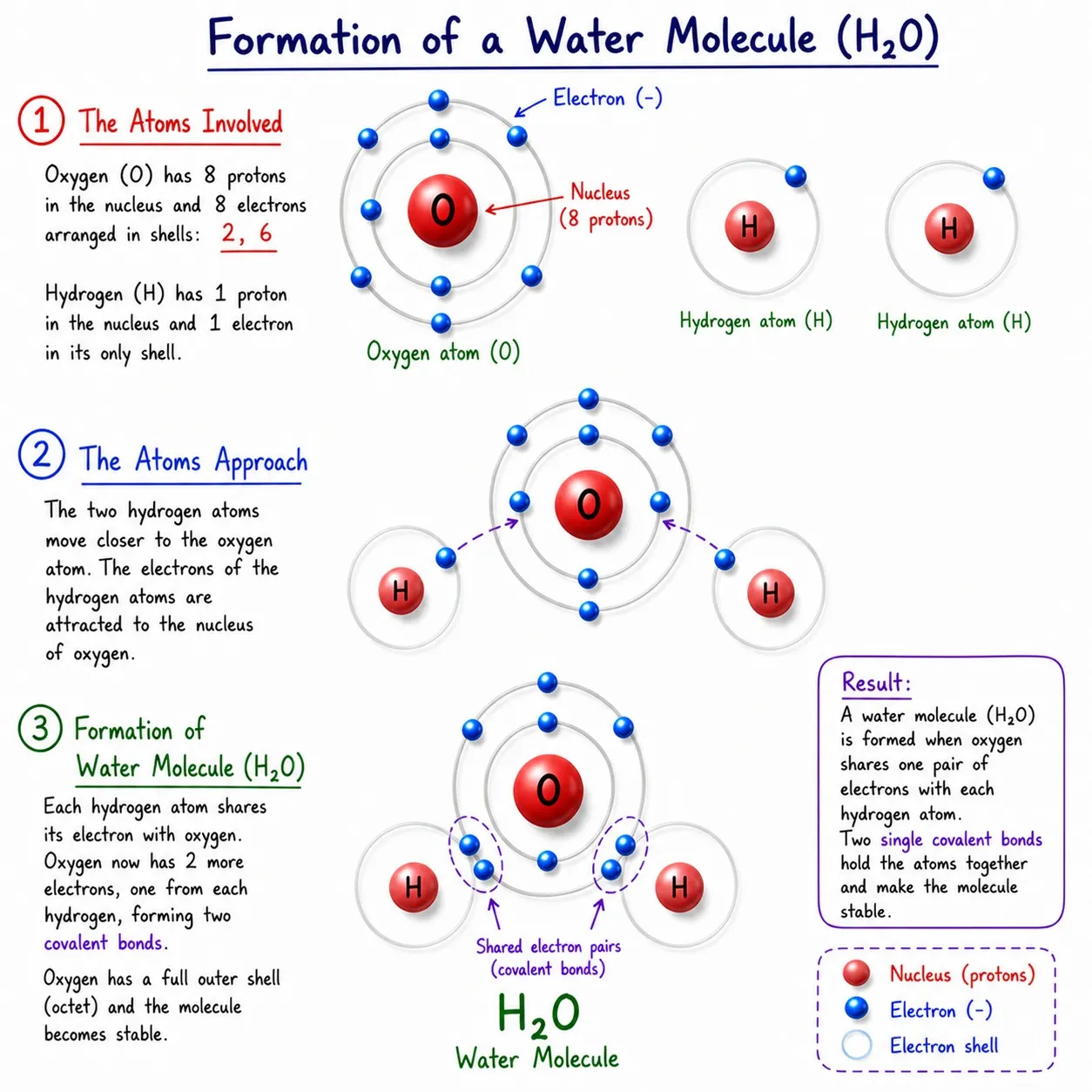
Water (H₂O)
➽ O ➜ 6 valence electrons, needs 2 more
➽ H ➜ needs 1 electron (only 1 H can’t fulfill O’s need)
➽ Solution: 2 H atoms, each shares 1 electron with O
➽ O gets 2 electrons, each H gets 1 ➜ all stable
➽ Represented as H₂O
Main Points
- Different element atoms can form covalent bonds by sharing electrons
- Each atom contributes electrons to complete its own valence shell
- When one atom needs 2 electrons ➜ 2 atoms of the other element may be required
- All such compounds formed by electron sharing = covalent compounds
C. Naming covalent compounds
1. Rules
➽ First element ➜ keeps its regular name
➽ Second element ➜ ends in -ide
➽ Use prefixes to indicate the number of atoms
➽ mono- is skipped for the first element, but used for the second
2. Prefixes
➽ 1-mono
➽ 2-di
➽ 3-tri, 4-tetra
➽ 5-penta
➽ 6-hexa
3. Vowel Rules
➽ Prefix ends in ‘o’ or ‘a’ + element starts with vowel ➜ drop last vowel
↪ mono + oxide ➜ monoxide (not monooxide)
↪ tetra + oxide ➜ tetroxide (not tetraoxide)
➽ Prefix ends in ‘i’ ➜ keep it
➽ di + oxide ➜ dioxide ✓
Special Cases
Hydrogen as the first element ➜ no prefix, regardless of atom count
- H₂S ➜ hydrogen sulfide (not dihydrogen sulfide)
Some compounds use only common names
- H₂O ➜ water (not hydrogen monoxide)
- NH₃ ➜ ammonia (not nitrogen trihydride)
Examples
| Formula | Name |
|---|---|
| CO | Carbon monoxide |
| CO₂ | Carbon dioxide |
| CS₂ | Carbon disulfide |
| PCl₃ | Phosphorus trichloride |
| SF₆ | Sulfur hexafluoride |
| N₂O₄ | Dinitrogen tetroxide |
| N₂O₅ | Dinitrogen pentoxide |
Bonding by electron transfer — Ionic bond
“The electrostatic force of attraction between oppositely charged ions that holds them together is called an ionic bond.“
When Does Electron Transfer Happen?
➽ Less than 4 valence electrons ➜atom tends to donate electrons
➽ More than 4 valence electrons ➜ an atom tends to gain electrons
Ions
Cation
formed by losing electrons ➜ positively charged
Anion
formed by gaining electrons ➜ negatively charged
Cations + Anions = collectively called Ions
Formation of NaCl
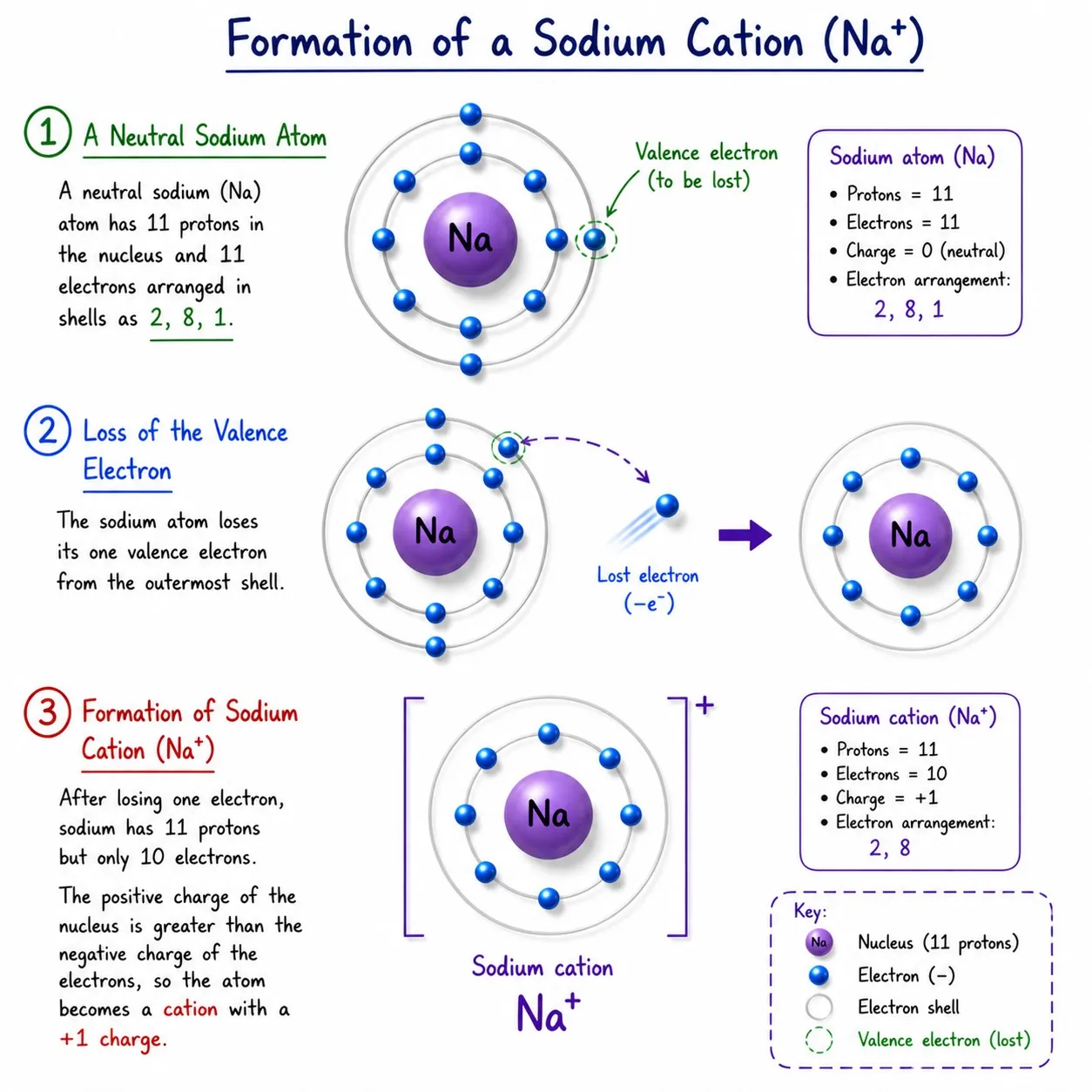
Sodium (Na) — atomic number 11
➽ Valence electrons: 1 ➜ loses it easily
➽ After losing 1e⁻ ➜ 11 protons, 10 electrons ➜ Na⁺ (cation)
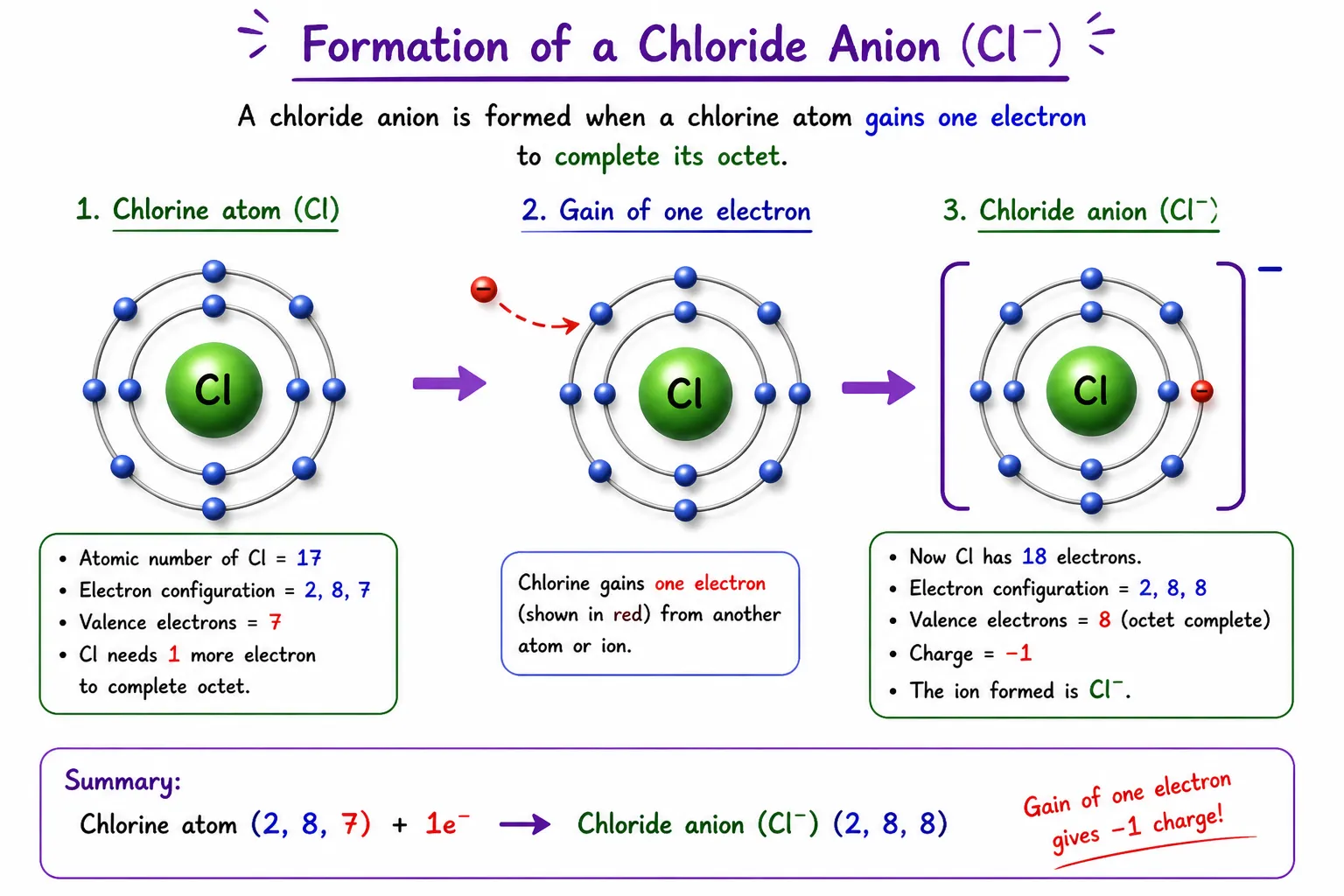
Chlorine (Cl) — atomic number 17
➽ Valence electrons: 7 ➜ needs 1 more
➽ After gaining 1e⁻ ➜ acquires negative charge ➜ Cl⁻ (anion)
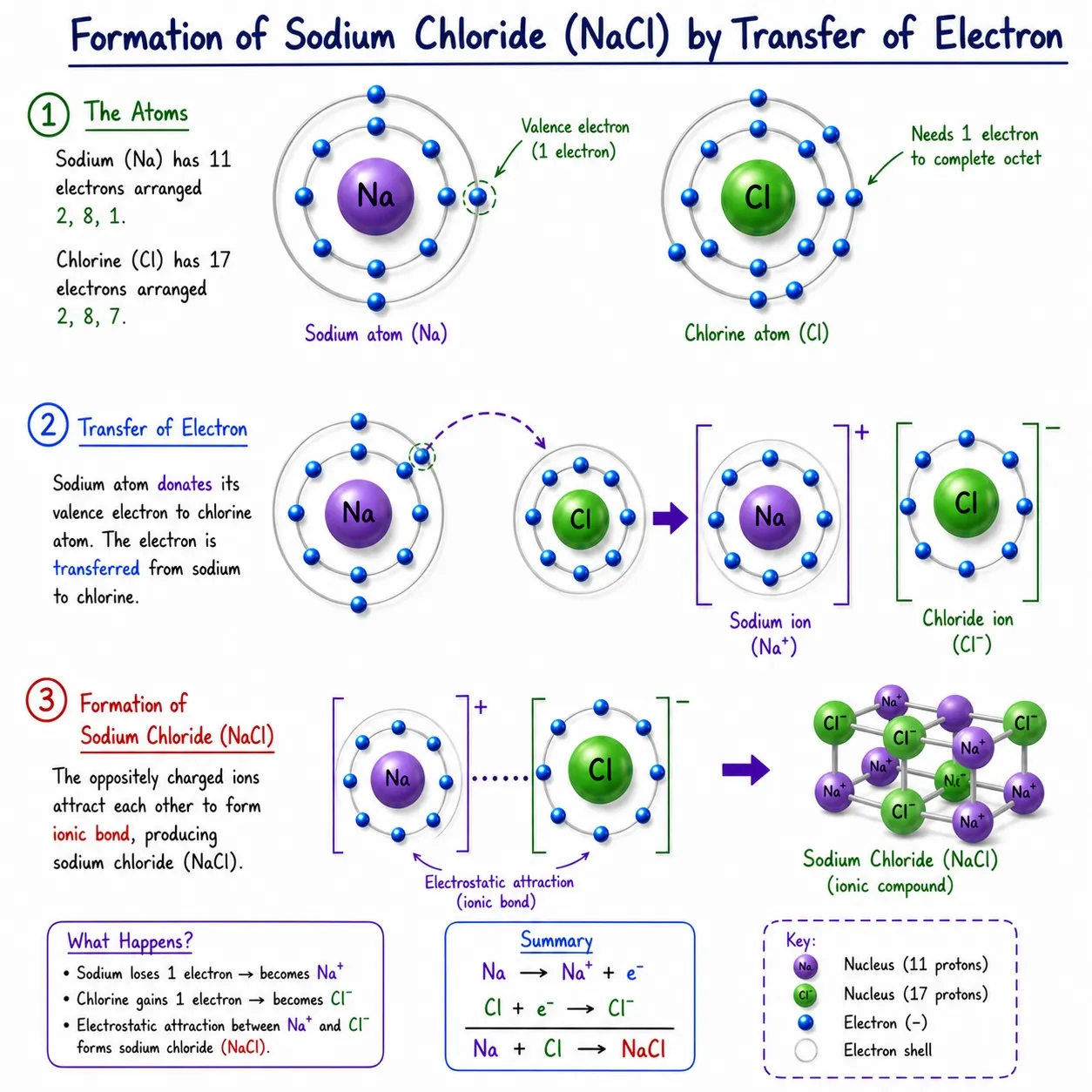
Na⁺ + Cl⁻ ➜ NaCl
Opposite charges attract ➜ ionic bond formed
Sulfur (S)
- Valence electrons: 6 ➜ needs 2 more to complete its octet
- Gains 2e⁻ ➜ S²⁻
Naming ionic compounds
Rules
- Cation name first, then anion name
- Simple anions end in -ide (e.g., chloride, oxide)
- Metals ➜ usually form cations
- Non-metals ➜ usually form anions
- Ionic compounds = typically metal + non-metal
- e.g., sodium chloride, calcium oxide, magnesium sulfide
Polyatomic Ions
- Formed by two or more elements combined
- Generally do not end in -ide
- e.g., sulfate, nitrate, hydroxide
Common Monoatomic Ions
Here is the information you provided, organized into a table:
| Name of ion | Formula | Valency |
|---|---|---|
| Sodium | Na⁺ | 1 |
| Lithium | Li⁺ | 1 |
| Potassium | K⁺ | 1 |
| Silver | Ag⁺ | 1 |
| Calcium | Ca²⁺ | 2 |
| Barium | Ba²⁺ | 2 |
| Iron (Ferrous) | Fe²⁺ | 2 |
| Iron (Ferric) | Fe³⁺ | 3 |
| Copper (Cuprous) | Cu⁺ | 1 |
| Copper (Cupric) | Cu²⁺ | 2 |
| Magnesium | Mg²⁺ | 2 |
| Zinc | Zn²⁺ | 2 |
| Aluminium | Al³⁺ | 3 |
| Fluoride | F⁻ | 1 |
| Chloride | Cl⁻ | 1 |
| Bromide | Br⁻ | 1 |
| Iodide | I⁻ | 1 |
| Oxide | O²⁻ | 2 |
| Sulfide | S²⁻ | 2 |
Common Polyatomic Ions
| Ion | Formula | Valency |
|---|---|---|
| Ammonium | NH₄⁺ | 1 |
| Hydroxide | OH⁻ | 1 |
| Nitrate | NO₃⁻ | 1 |
| Hydrogencarbonate | HCO₃⁻ | 1 |
| Carbonate | CO₃²⁻ | 2 |
| Sulfate | SO₄²⁻ | 2 |
Writing Chemical Formulae
Two Ways to Write Formulae
- Based on electrons shared/transferred
- Crossover method (valency method) ➜ quicker
Writing chemical formulae of covalent compounds
➽ Write symbols of the elements
➽ Write their valencies
➽ Crossover the valencies ➜ write as subscripts after each symbol
Crossover Method (How it works)
➽ Valency of A becomes a subscript of B, and vice versa
➽ If both valencies are equal ➜ subscript is 1 (write just the symbol)
➽ If the ratio can be simplified ➜ simplify it
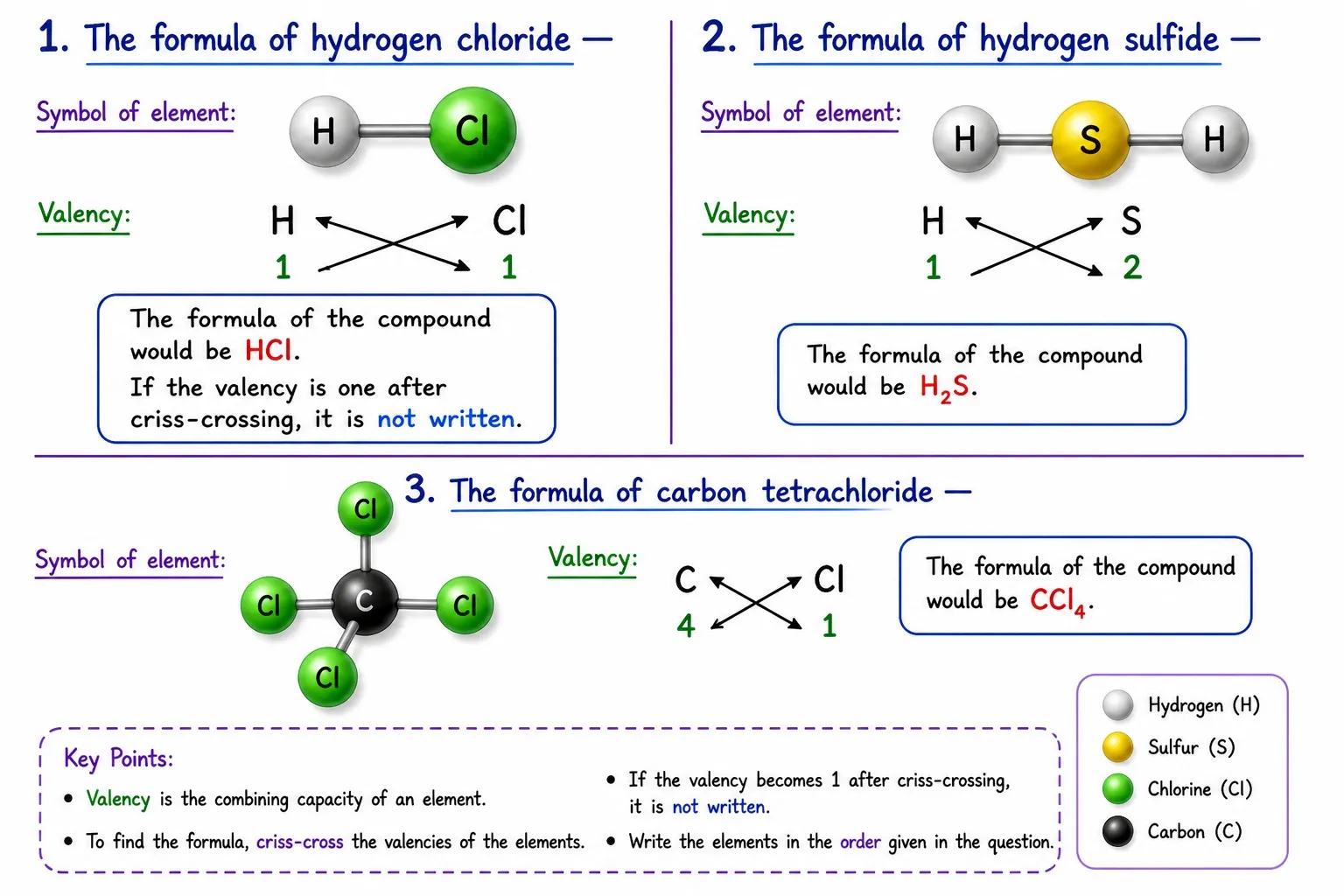
Important Points
➽ Always simplify the ratio if possible
➽ Subscript of 1 is not written explicitly
➽ Polyatomic ions in brackets if subscript > 1
e.g., Ca(OH)₂ — not CaOH₂
Writing chemical formulae of ionic compounds
Steps
➽ Write the cation symbol first, then the anion
➽ Write charges below the symbols (not as superscripts)
➽ Crossover only the numbers of the charges as subscripts
➽ Simplify the ratio if possible (divide by a common factor)
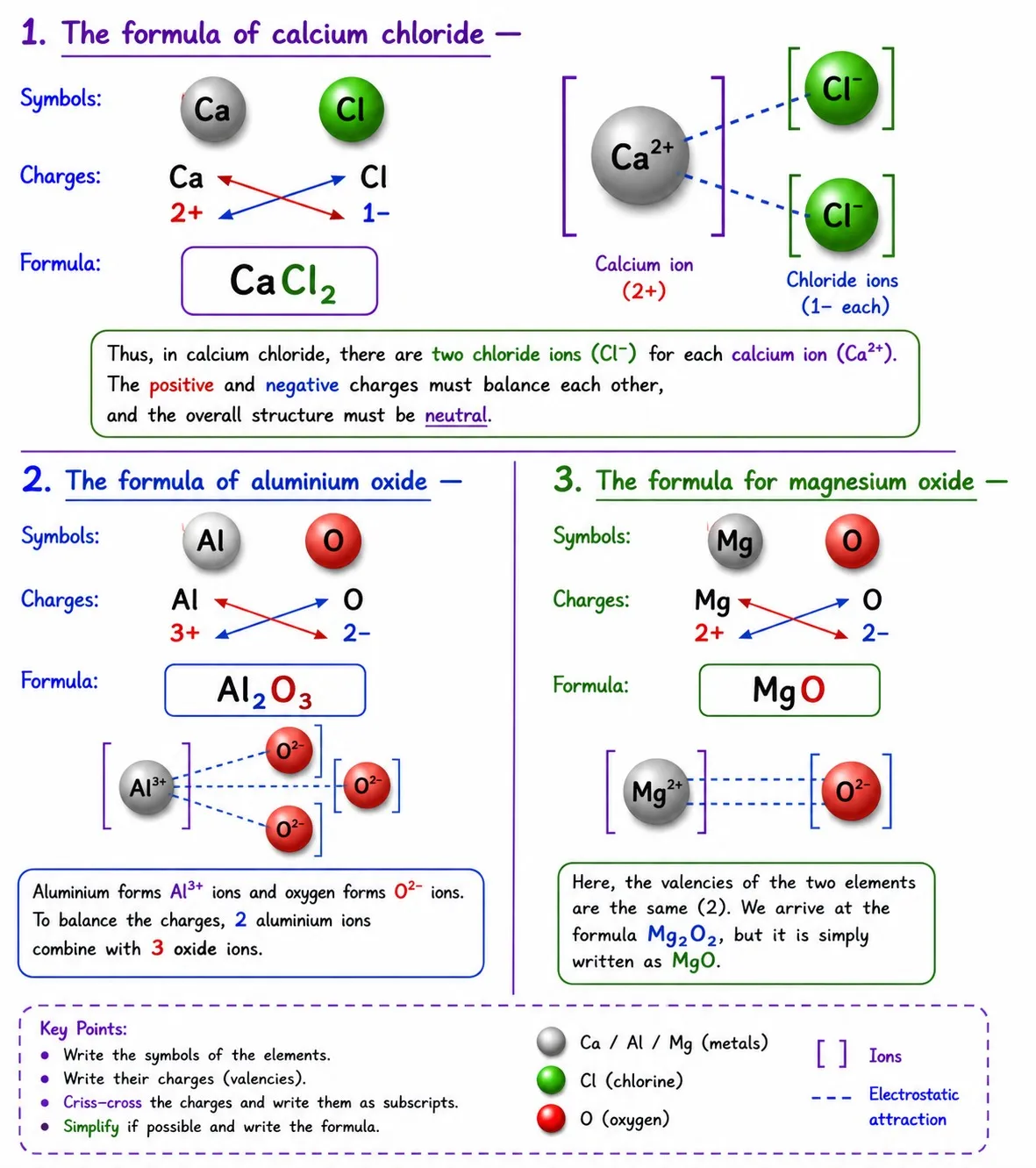
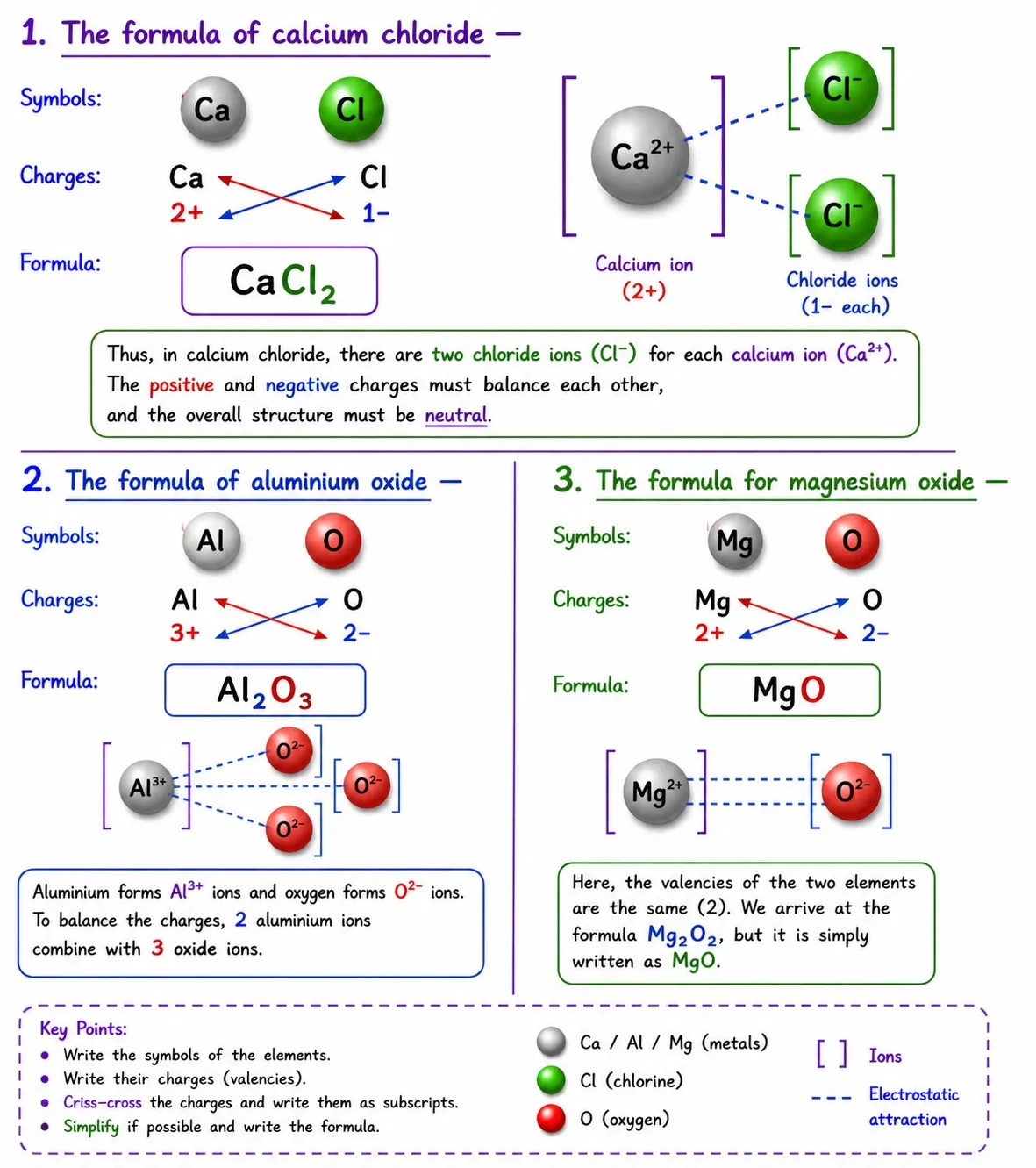
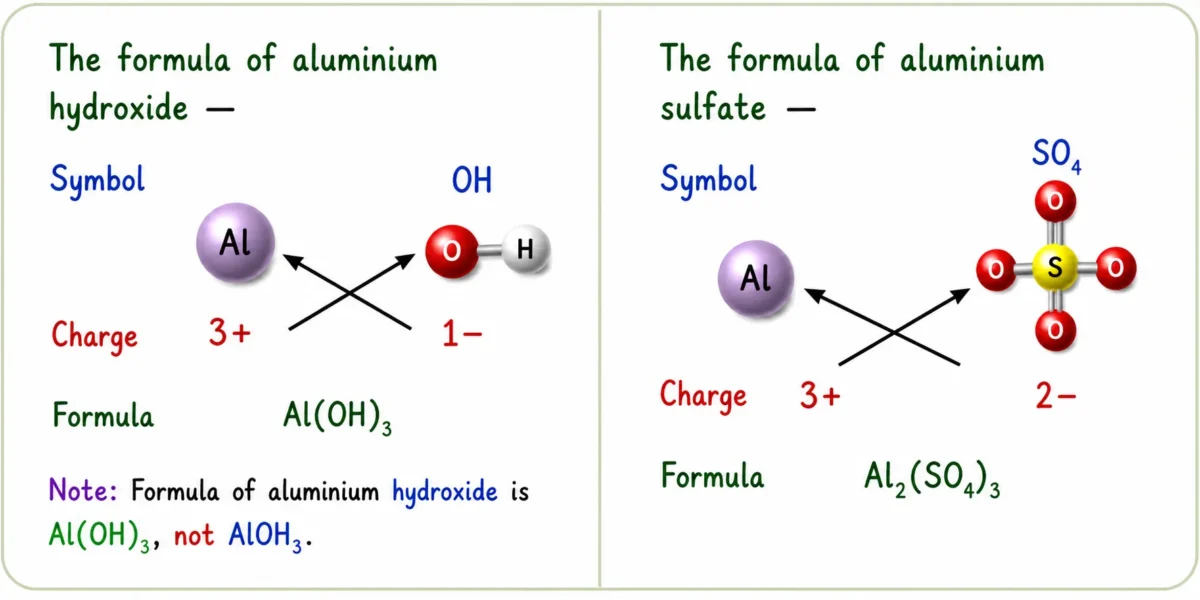
hydroxide|Ch 9 Atomic Foundations of Matter Notes Class 9
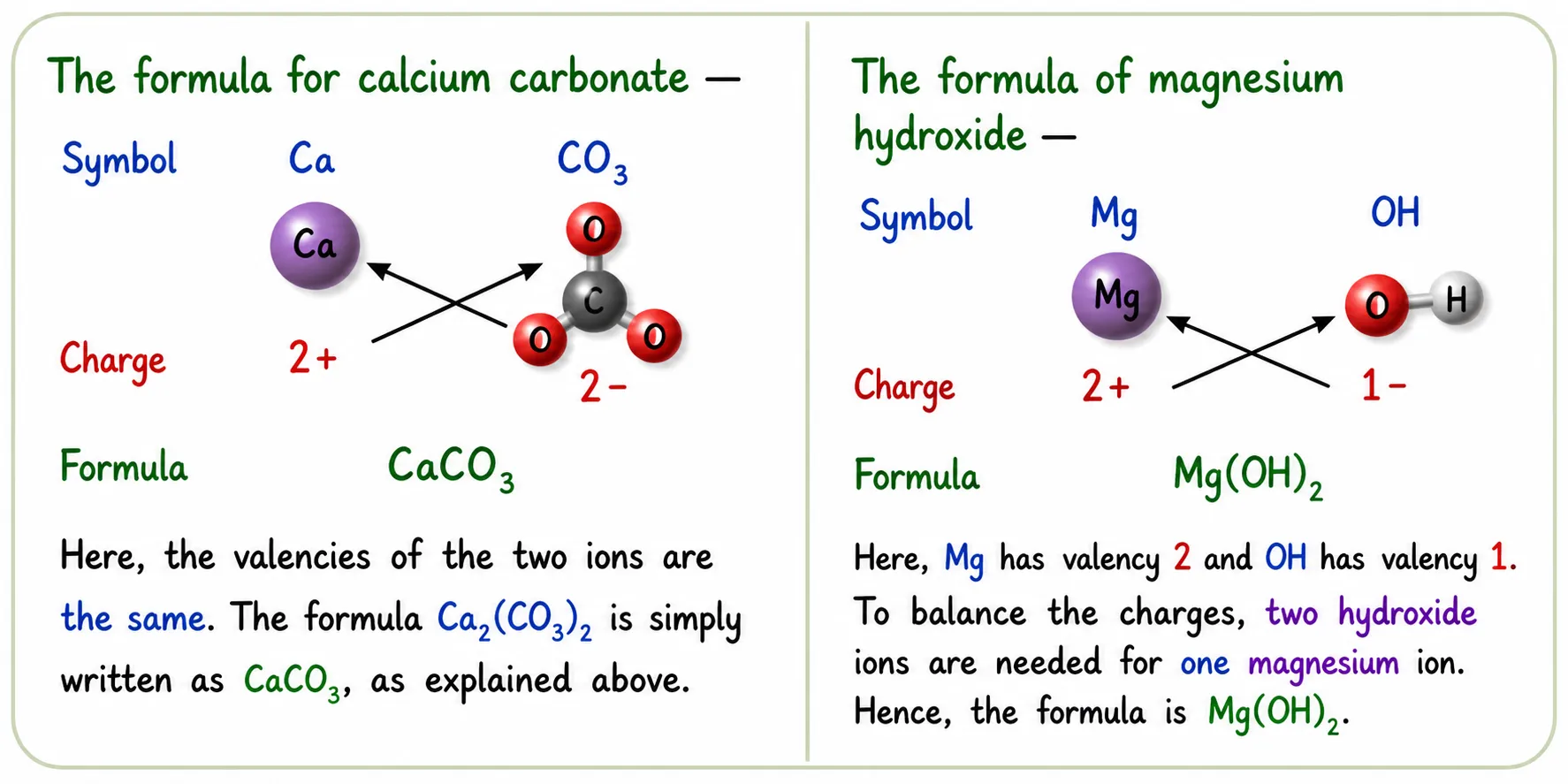
Polyatomic Ions — Use Brackets
➽ Use ( ) when 2 or more of the same polyatomic ions are present
➽ No brackets needed if only one polyatomic ion
Properties of the Ionic and the Covalent
Compounds
Solubility
| Property | Ionic | Covalent |
|---|---|---|
| In water | ✓ Soluble | ✗ Mostly insoluble |
| In kerosene/petrol | ✗ Insoluble | ✓ Soluble |
Rule of thumb: “Like dissolves like”
1. Electrical Conductivity
Ionic Compounds
Solid state ➜ do NOT conduct
➽ Because Ions held in fixed positions cannot move
Dissolved in water ➜ conduct electricity
➽ Ions become free to move in solution
Covalent Compounds
Do NOT conduct in any state
Even if soluble in water (e.g., sugar) ➜ no ions formed in solution ➜ no conductivity
Camphor, naphthalene ➜ insoluble + no ions ➜ no conductivity
2. Melting & Boiling Points
Ionic ➜ high melting & boiling points
➽ Strong inter-ionic attractions hold them together
Covalent ➜ low melting & boiling points
Molecular Mass of Covalent Compounds
“Molecular Mass = sum of atomic masses of all atoms in the molecule“
How to Calculate
- Write the formula
- Note the atomic mass of each element
- Multiply atomic mass × number of atoms
- Add all values
Examples
Water (H₂O)
- H = 1 u, O = 16 u
- (1 × 2) + (16 × 1) = 18 u
Carbon Dioxide
(CO₂)
- C = 12 u, O = 16 u
- (12 × 1) + (16 × 2) = 44 u
Important Note — Ionic Compounds
➽ Ionic compounds form 3D crystals, not molecules
➽ Therefore, ionic compounds do not have a molecular mass
➽ Instead, their mass is expressed as formula mass
Formula Unit Mass of Ionic Compounds
Formula unit:
A collection of the simplest whole-number ratios of ions in an ionic compound
Formula unit mass:
Mass of one formula unit (calculated the same way as molecular mass)
How to Calculate
- Write the formula
- Note atomic masses
- Multiply atomic mass × number of atoms
- Add all values
- For polyatomic ions ➜ calculate ion mass first, then multiply by the subscript
Examples
Sodium Oxide
(Na₂O)
- Na = 23 u, O = 16 u
- (23 × 2) + (16 × 1) = 62 u
Calcium Nitrate
Ca(NO₃)₂
- Ca = 40 u, N = 14 u, O = 16 u
- Ca: 40 × 1 = 40
- NO₃ group:
(14 × 1) + (16 × 3) = 62 → × 2 = 124 - 40 + 124 = 164 u


![[New Book] How Forces Affect Motion Notes Ch 6](https://studyless.in/wp-content/uploads/2026/04/How-Forces-Affect-Motion-Notes-Ch-6-2-1200x675.jpg)

