You know, matter, now it is the time to know what actually a matter is made of.
Particulate Nature of Matter Class 8 Chapter 7 takes you deep into the concept of matter. So, there will be new keywords and concepts to keep in mind.
And hence, we have created Particulate Nature of Matter Short Notes Class 8 to give you all the keywords and concepts in an easy-to-learn, memorise, and revise format. Read and understand your NCERT textbook.
Then, revise from the short notes below again and again to master Chapter 7 of the Science of Curiosity book.
The PDF of “Particulate Nature of Matter Short Notes Class 8” is at the end of the page.
Happy learning!
What Is Matter Composed of?
Composition of Matter
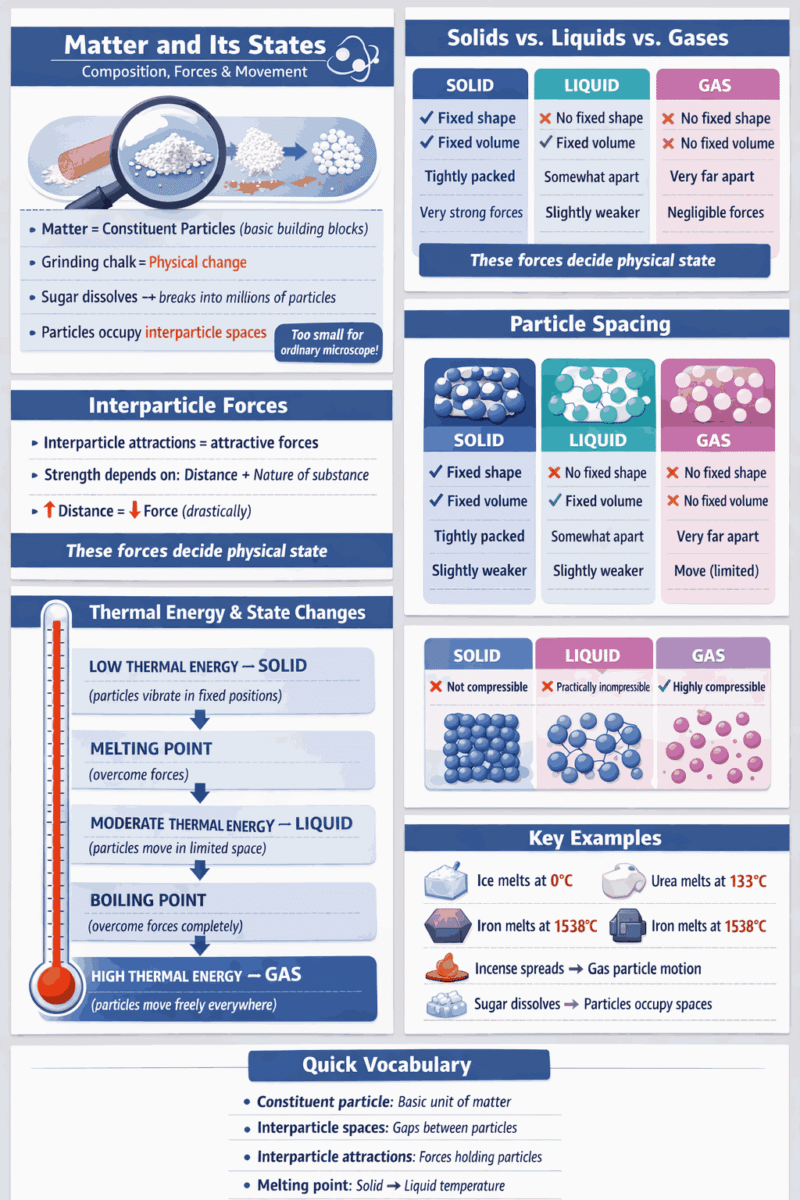
Matter is composed of extremely small constituent particles – the basic building blocks of substances.
Grinding chalk = physical change (no new substance formed, only size reduction)
Constituent particles cannot be broken down further.
Sugar in Water Example
1. Sugar dissolves in water → breaks into constituent particles
2. Particles not visible but present (sensed by taste)
3. One sugar grain = millions of constituent particles
4. Sugar particles occupy the interparticle spaces between water particles
Key fact:
Particles cannot be seen through an ordinary microscope
Interparticle Forces
Interparticle attractions = attractive forces holding particles together
Strength depends on:
1. Nature of substance
2. Interparticle distance
(slight increase → drastic decrease in force)
Interparticle attractions decide the physical state of matter.
Three States of Matter
Solid State: Properties and Particle Behavior
Properties:
- Definite shape
- Definite volume
Particle behavior:
- Tightly packed
- Very strong interparticle attractions
- Fixed positions
- Only vibrate/oscillate
(cannot move past each other)
On heating:
1. Vibrations increase
2. Particles leave positions
3. Interparticle forces weaken
4. Converts to liquid
Melting point
minimum temperature for solid to liquid conversion at atmospheric pressure
| Material | Melting Point |
|---|---|
| Ice | 0 °C |
| Urea | 133 °C |
| Iron | 1538 °C |
◾ Weak interparticle forces = low melting point.
◾ Strong interparticle forces = high melting point
Liquid State: Properties and Particle Behavior
Properties:
- No fixed shape (takes container shape)
- Definite volume
Particle behavior:
- Free to move within a limited space
- Somewhat farther apart
- Interparticle attractions = slightly weaker
than solids
(but strong enough to keep particles close)
On heating:
1. Particles move vigorously
2. Move apart
3. Interparticle forces decrease
4. Converts to vapour/gas
Boiling point
The temperature at which a liquid changes to a vapour at atmospheric pressure
At boiling
point:
- Vapour formation = very fast
- Occurs at the surface + within the liquid
- Visible as bubble formation
Below
boiling point:
- Vapour formation = slow
- Occurs only at the surface
- Called evaporation
Gaseous States Properties and Particle Behavior
Properties:
- No fixed shape
- No fixed volume
Particle behavior:
- Move freely in all directions
- Negligible interparticle attractions
Fluids
liquids + gases (both flow, no fixed shape)
State Comparison
| Property | Solid | Liquid | Gas |
|---|---|---|---|
| Shape | Fixed | Not fixed | Not fixed |
| Volume | Fixed | Fixed | Not fixed |
| Packing | Tightly packed | Somewhat apart | Very far apart |
| Interparticle Force | Very strong | Slightly weaker | Negligible |
| Movement | Vibrate in fixed positions | Free movement (limited space) | Free movement (all directions) |
Key Terms
◼Interparticle spaces – Gaps between particles
◼Interparticle attractions – Attractive forces holding particles together
◼Interparticle distance – Distance between particles
◼Evaporation – Slow vapour formation below boiling point (surface only)
◼Fluids – Liquids + gases (flow, no fixed shape)
◼Vibrate/Oscillate – Move to and fro about a fixed position
How Does the Interparticle Spacing Differ in the Three States of Matter?
Compressibility
Gas
(Air)
- Gas particles have a lot of space between them
- Space can be reduced by external pressure
- Stop pushing → particles spread
Liquid
(Water)
- Water is practically incompressible
Dissolution of Sugar
Observation:
- Initially, the water level increases when sugar is added
- After dissolution, the level may decrease to some extent
- Volume of solution < sum of water + sugar volumes
Conclusion:
Space exists between water particles → dissolved sugar particles occupy these spaces
Sand in Water
- Sand = a solid that does not dissolve
- Sand particles settle down
- Occupy space in the container
- Total volume increases
Interparticle Spacing in Solids
- Particles held by strong forces of attraction
- Do not move from place to place
- Closely packed
- Some space left between particles (despite close packing)
- Space between particles ≠ of air
- Contains nothing at all
Interparticle Spacing Summary
| State | Particle Packing | Interparticle Space | Compressibility |
|---|---|---|---|
| Solid | Closely packed | Minimum space (some exists) | Not compressible |
| Liquid | Less closely packed | More space than solids | Practically incompressible |
| Gas | Very loosely packed | Maximum space (lot of space) | Highly compressible |
How Particles Move in Different States
of Matter?
Particle Motion in Liquids
Potassium permanganate in water:
① Water particles are in constant motion
② Water particles pull out particles from the potassium permanganate grain
③ Water particles hit these particles
④ Particles spread throughout the liquid
Insoluble
substances
(like sand):
- Constituent particles held together strongly
- Water particles unable to pull these out
- Remain insoluble
Particle Motion in Gases
Incense stick experiment:
- Fragrance is first noticed near the incense stick.
- Soon, it spreads across the whole room.
- This happens because air particles are always moving.
- They collide with fragrance particles and carry them everywhere.
Thermal Energy and States of Matter
Key principle:
The thermal (heat) energy of particles determines the physical state of matter.
Relationship:
- Thermal energy determines the distance between particles
- The distance between particles determines the strength of attractive forces
Solid State
Thermal Energy
- Low thermal energy
- Particles remain close to each other
- Strong interparticle attractive forces
- Motion restricted to small vibrations only
At the
melting point:
- Thermal energy is used to
overcome attractive forces - Solid → liquid
- Particles move away from fixed positions
- Interparticle distance increases slightly
- Strength of attractive forces reduces
- Particles can move around
(within a limited space)
Liquid State
Thermal energy
- Moderate thermal energy
- Particles can move around
- Movement within a limited space
- Interparticle distance = slightly more than solids
- Attractive forces = reduced but still present
Gaseous State
Thermal Energy
- High thermal energy
- Particles have enough energy
to overcome the forces of attraction - Move freely in all directions
Particle Nature Summary
| State | Thermal Energy | Interparticle Distance | Attractive Forces | Particle Motion |
|---|---|---|---|---|
| Solid | Low | Minimum | Strong | Small vibrations only |
| Liquid | Moderate | Slightly increased | Reduced (still present) | Move around (limited space) |
| Gas | High | Maximum | Overcome (negligible) | Move freely (all directions) |
FAQs: Particulate Nature of Matter Short Notes Class 8
1. Why can we compress air in a syringe but not water?
Gas particles naturally have a lot of space between them, which can be reduced by applying external pressure. When you push the plunger, the particles are forced closer together, decreasing the volume. Water is practically incompressible because liquid particles are already closely packed with minimal interparticle space.
2. What happens to sugar particles when sugar dissolves in water?
Sugar breaks into constituent particles that cannot be broken down further. These particles occupy interparticle spaces between water particles. Water particles are in constant motion – they pull out sugar particles from grains and hit them to spread throughout the liquid. Sugar becomes invisible but remains present (sensed by taste).
3. Why do solids have high melting points while some have low melting points?
Melting point depends on the strength of interparticle forces. Solids with weak interparticle attractions have low melting points (e.g., ice = 0°C). Solids with strong attractive forces have high melting points (e.g., iron = 1538°C). The stronger the forces holding particles together, the more thermal energy is needed to overcome them.
4. How does thermal energy determine the state of matter?
Thermal (heat) energy determines the distance between particles, which determines the strength of attractive forces. Solid state = low thermal energy → particles close → strong forces → small vibrations only. Liquid state = moderate thermal energy → slightly increased distance → reduced forces → movement within a limited space. Gaseous state = high thermal energy → particles overcome forces → free movement in all directions.
5. What is the difference between evaporation and boiling?
Boiling occurs at the boiling point – vapour formation is very fast, happens at surface + within liquid, visible as bubble formation. Evaporation occurs at all temperatures below boiling point – vapour formation is slow, occurs only at surface. Both convert liquid to vapour, but speed and location differ.
6. Why does the fragrance from an incense stick spread throughout the room?
Gas particles are in constant motion. Initially, fragrance is felt only around incense stick. Air particles moving constantly hit the fragrance particles and help them spread throughout the room. Gas particles have negligible interparticle attractions and move freely in all directions, allowing fragrance to fill entire space.
7. Why does grinding chalk only cause physical change and not chemical change?
Grinding chalk does not change it into a new substance – it remains chalk. Only the size of each speck reduces. Even after grinding to smallest possible size, you reach constituent particles that cannot be broken down further. Since no new substance forms and only physical appearance (size) changes, it is a physical change.
8. What happens to interparticle forces when a solid is heated to its melting point?
When solids are heated, particles vibrate more vigorously. At melting point, vibrations become so vigorous that particles start leaving their fixed positions. Interparticle forces of attraction weaken, and solid converts to liquid state. Thermal energy is used to overcome attractive forces between particles.
9. Why can you move your finger through water but not through a solid?
In liquids, interparticle attractions are slightly weaker than solids, but strong enough to keep particles close. When you move finger through water, you temporarily displace it. Position is restored when finger is removed. In solids, particles are in fixed positions with very strong interparticle attractions – they cannot be displaced without breaking/cutting permanently.
10. Why is the volume of the sugar solution less than the sum of the water and sugar volumes?
Water particles have interparticle spaces (gaps) between them. When sugar dissolves, sugar particles occupy these spaces instead of adding to total volume. Volume of solution is less than sum of water + sugar volumes because dissolved particles fit into existing spaces between water particles. This proves that space exists between particles of matter.
PDF: Particulate Nature of Matter Short Notes Class 8 Chapter 7




