Short Notes: Electricity — Magnetic and Heating Effects (Class 8)
These short notes are clear, concise, and exam-ready. They are designed for quick revision.
The notes are based on Curiosity — Textbook of Science for Grade 8.
First, study the chapter from your textbook. Then use these notes to revise the key ideas.
Your textbook includes many important activities. These activities help you understand the concepts better. They also make the ideas easier to remember for exams.
Now, let’s begin.
Does An Electric Current Have A Magnetic Effect?
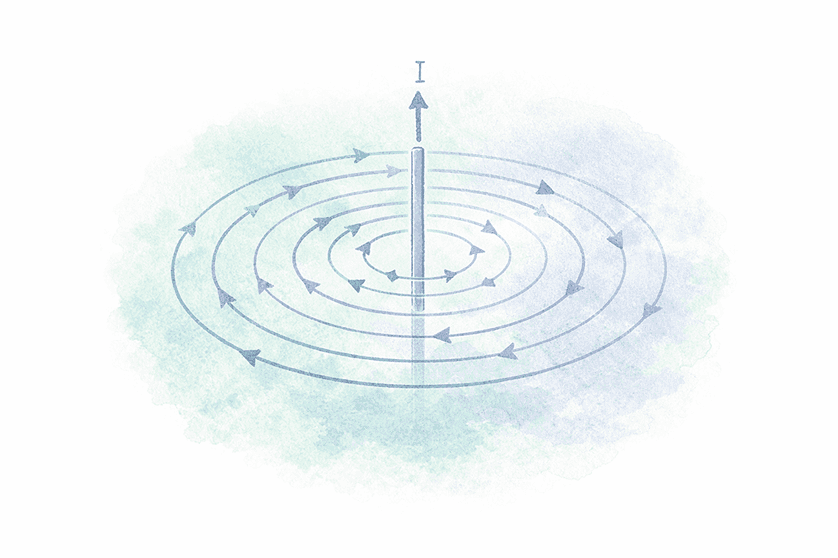
📌 Key Observation
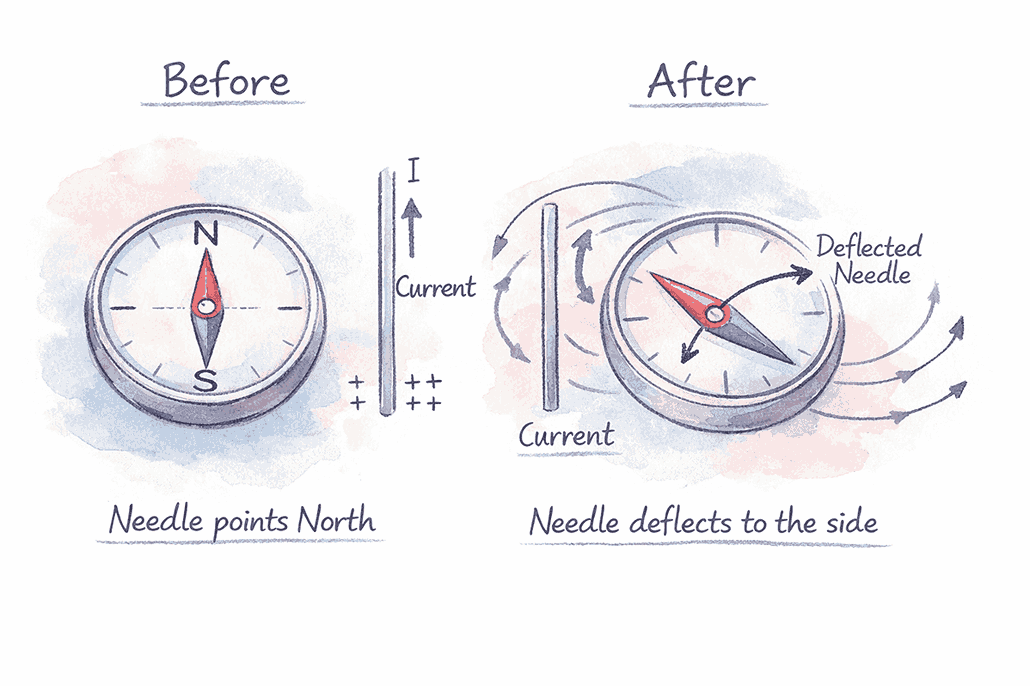
Compass Needle Behaviour:
- ✓ When current flows → Compass needle deflects from original direction
- ✓ When the current stops → Needle returns to the original direction
🔍 Why Does This Happen?
Understanding the Compass:
- Compass needle = tiny magnet
- Deflects when a magnet is brought near it
- Magnetic effect can act through non-magnetic materials
The Connection:
- Deflection indicates: Current-carrying wire has magnetic effect on compass needle
- When current stops → Magnetic effect disappears
🌐 Magnetic Field
Definition:
The region around a magnet or current-carrying wire where its magnetic effect can be felt (such as by compass needle deflection)
Key Points:
- A magnetic field exists when current flows
- The magnetic field disappears when the current stops
🧲 Electromagnets
🔍 What is an Electromagnet?
Observation:
- When current passes through a cylindrical coil → behaves like a magnet → deflects the compass needle
- When an iron nail is inserted in the core of the coil:
- The coil becomes a stronger magnet
- Deflection of the compass needle is much more
- Attracts iron clips
- When the current is stopped → coil loses its magnetic effect
Definition:
A current carrying coil that behaves as a magnet is called an electromagnet
🧭 Polarity of Electromagnets
Basic Rule:
- When two magnets are brought close, unlike poles (North–South) attract each other
Finding Polarity:
- If the north pole of the magnetic compass is attracted towards end A of the electromagnet → end A is the south pole
- Repeat the procedure to find the polarity of end B
- The polarity of end B is opposite to the polarity of end A
Conclusion:
| Feature | Detail |
|---|---|
| Poles in electromagnet | Two — North and South |
| Similar to | Poles in an electromagnet |
🏗️ Lifting Electromagnets
What are they?
- Strong electromagnets that may be hung on cranes
How they work:
| Current State | Action |
|---|---|
| ✅ Current ON | Electromagnet lifts iron/steel objects |
| ❌ Current OFF | Magnetic field disappears → objects released |
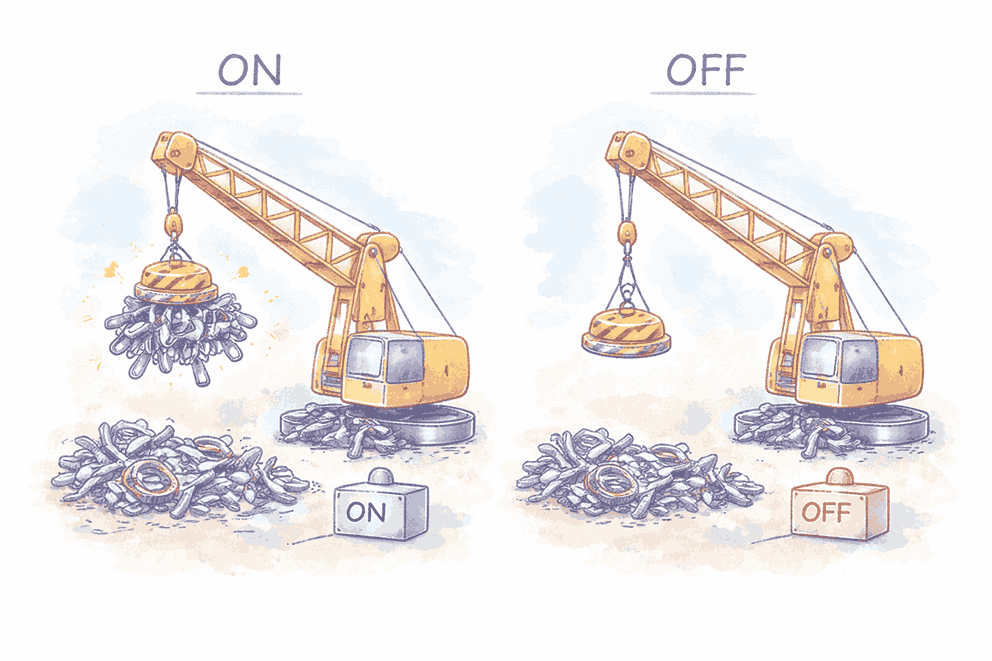
Applications
- Used in factories and scrap yards
- Purpose: Move, lift, and sort heavy metal items efficiently
Does a Current-Carrying Wire Get Hot?
📌 Key Observation
Nichrome wire feels warm when current is passed through it
🔍 Why Does This Happen?
Resistance in Conductors:
- When electric current flows through any conductor → faces opposition or resistance to its flow
- Different conductors offer different levels of resistance
| Conductor | Resistance Level |
|---|---|
| Nichrome wire | Higher resistance |
| Copper wire (same size & length) | Lower resistance |
Energy Conversion:
- Resistance causes electrical energy → heat energy
Definition:
When current passes through a conductor → it gets heated. This warming is known as the heating effect of electric current
Everyday Applications Of The Heating Effect Of Electric Current
How an Incandescent Lamp Works:
Filament is heated by an electric current → lamp glows
Household Appliances
Using the Heating Effect:
- Electric room heaters
- Stoves
- Irons
- Immersion rods
- Water heaters
- Kettles
- Hair dryers
Common Feature:
- All these devices contain a rod or coil of wire called a heating element
- In some appliances where the element is visible → it can be seen glowing red hot
Useful vs. Problematic
| Aspect | Details |
|---|---|
| ✅ Useful | In many everyday appliances |
| ❌ Energy loss | In wires during transmission |
| ❌ Overheating | May damage plugs & sockets; plastic parts may melt |
| ❌ Fire risk | Overheating may lead to fires |
| 🛡️ Safety | Household circuits have safety devices to minimise such incidents |
How Does a Battery Generate Electricity?
Voltaic Cell (Galvanic Cell)
Structure
- Two metal plates made of different materials (called electrodes)
- A liquid called an electrolyte (usually a weak acid or salt solution)
- Placed in a glass or plastic container
- Electrodes partly dipped in electrolyte
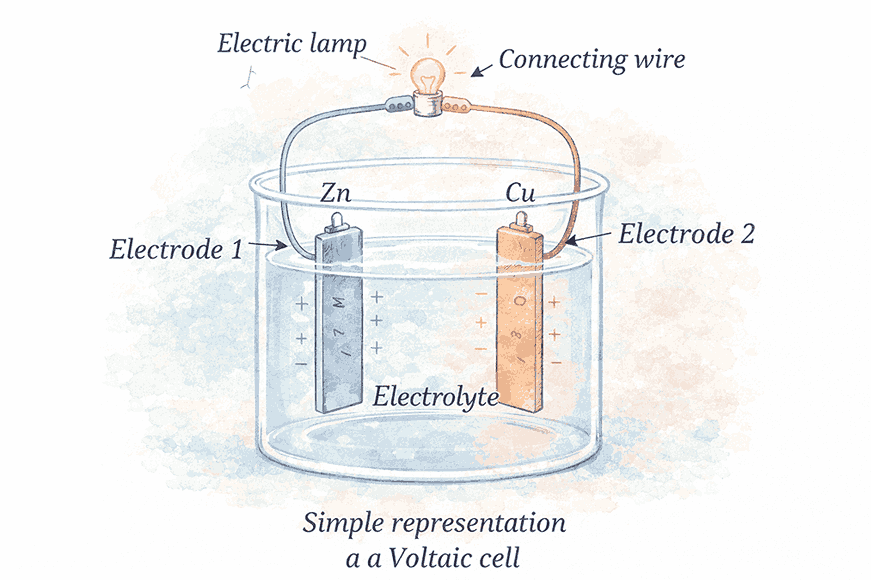
How it Works:
- Chemical reaction between plates and electrolyte → produces electricity
- When the circuit is connected, current flows from the positive terminal → through the circuit → to the negative terminal
Limitation:
| Stage | What Happens |
|---|---|
| ✅ Working | Chemicals produce electricity |
| ❌ Dead | Chemicals get used up → cell stops working → cannot supply any more electricity |
🍋 Lemon Cell Activity
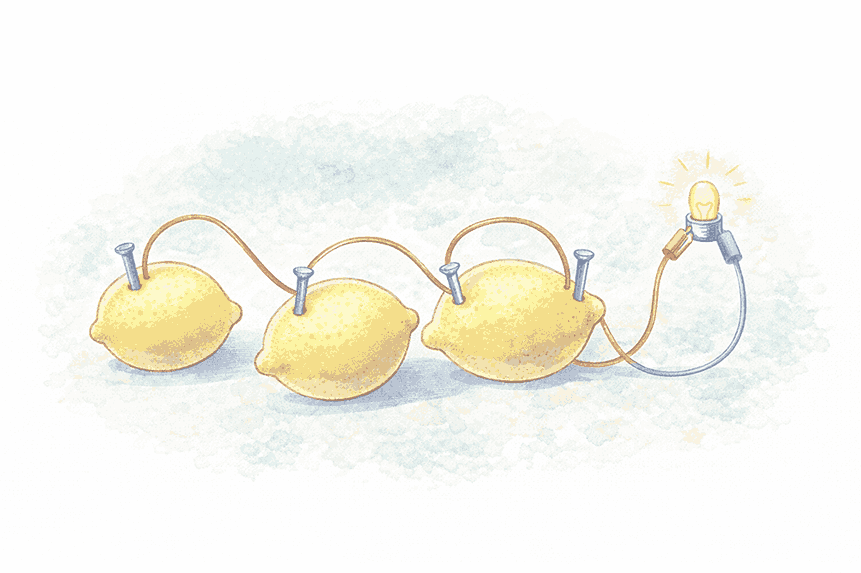
Materials Required
- 5–6 juicy lemons
- Copper wires/strips (1–2 mm thick)
- Iron nails
- 1 LED
- Connecting wires
Procedure
- Insert a copper wire and an iron nail into one lemon, keeping them apart by a small distance
- Repeat for all remaining lemons
- Join copper wires and nails as shown in the diagram
- Connect the LED between:
- Copper wire of the first lemon
- Iron nail of the last lemon
Observation
- ✅ LED glows → Cell is working
Key Components:
| Component | Material Used |
|---|---|
| Electrodes | Copper wires and iron nails |
| Electrolyte | Lemon juice (conducts electricity) |
| Alternative electrolyte | Salt solutions |
🔋 Dry Cells
Why Dry Cells?
- Voltaic cells not convenient for everyday use
- Dry cells = most widely used electric cells today
Why ‘Dry’?
- Electrolyte is not liquid but a thick, moist paste
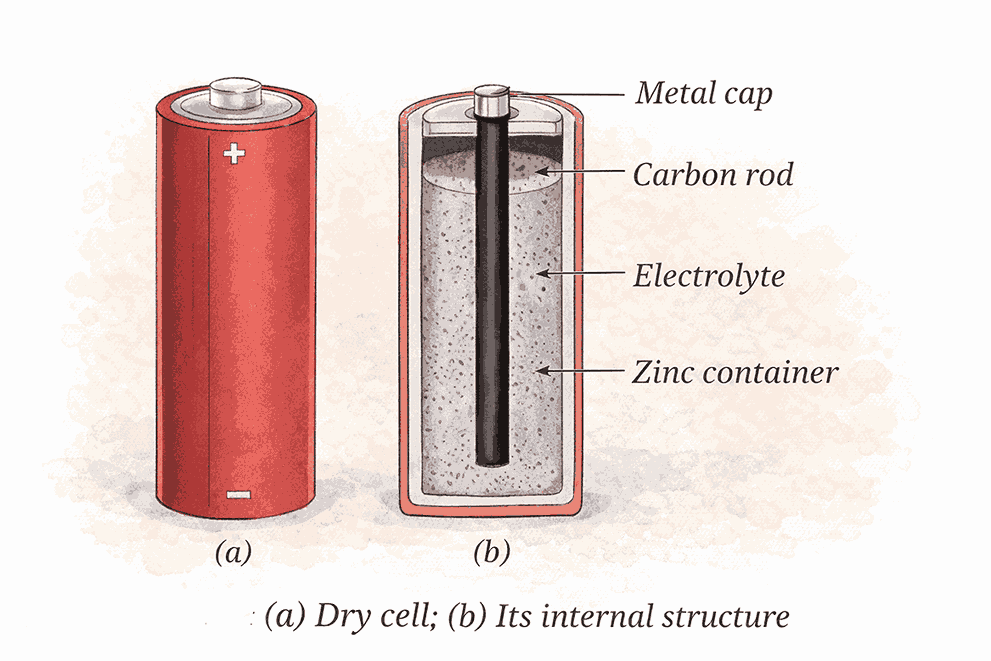
Structure:
| Part | Function |
|---|---|
| Zinc container | Acts as negative terminal |
| Carbon rod (centre) with metal cap | Surrounds the carbon rod |
| Paste-like electrolyte | Surrounds carbon rod |
Type:
- Single-use cell → once used up → must be disposed of.
🔁 Rechargeable Batteries
Advantages:
- Can be recharged and reused multiple times
- Prevents wastage
- Saves money over time
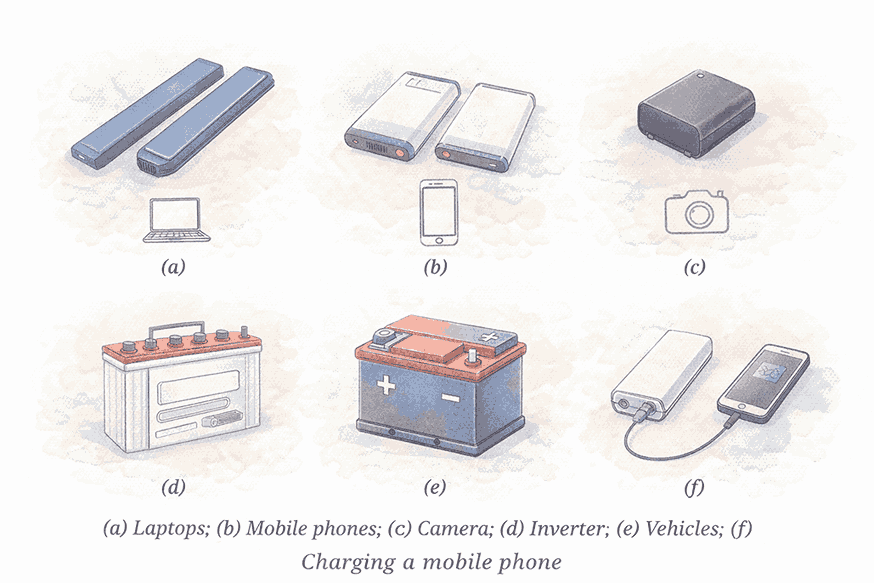
Applications by Size:
| Size | Used In |
|---|---|
| Small | Watches, phones |
| Medium | Laptops, tablets |
| Large | Inverters, electric vehicles |
Limitation:
- Do not last forever
- After being charged and used many times → slowly wear out
Also Read| Health: The Ultimate Treasure Short Notes Class 8 Chapter 3




