In the previous chapter of the book, Curiosity Grade 8, you studied the particulate nature of matter.
Now, in Nature of Matter: Elements, Compounds, and Mixtures (Chapter 8), you will study how matter exists.
Matter exists in different forms such as elements, compounds, and mixtures. After understanding this chapter, you need short notes that are clear, to the point, and easy to read, memorise, and revise.
And “Nature of Matter Elements Compounds And Mixture Notes” just does that. An easy-to-read and revise resource for you. Revise it again and again to get good marks in the exam.
The PDF of Notes for Chapter 8 – Nature of Matter, Elements, Compounds, and Mixtures is at the end of the page.
What Are Mixtures?
What Are
Mixtures?
- Mixture = two or more substances mixed
- Substance retains its own properties
- Substances do not react chemically
Examples:
poha, sprout salad, sugar dissolved in water, lemonade, soups
Types of Mixtures
| Feature | Non-Uniform Mixture | Uniform Mixture |
|---|---|---|
| Visibility | Components visible to the naked eye or a magnifier | Components not distinguishable, even with a microscope |
| Distribution | Uneven | Evenly distributed |
| Examples | Sprout salad, poha, soil | Sugar water, salt water, air |
Air: Uniform
Mixture
- Air is a uniform mixture of gases
- Main components:
| Gas | Approx. % | Key Role |
|---|---|---|
| Nitrogen | 78% | Do not support combustion |
| Oxygen | ~21% | Essential for life and combustion |
| Argon, CO₂, Water vapour | Trace | CO₂ detected via lime water test; water vapour condenses on cool surfaces |
Lime water test for CO₂:
Calcium hydroxide + Carbon dioxide → Calcium carbonate (white precipitate) + Water
→ Lime water turns milky when CO₂ is present
Dust in the air:
1. Tiny shining particles visible in sunlight beams
2. Not part of the air; considered pollutants
3. Amount varies by location and time
Scientific Definition of Mixture
- Scientific mixture: Only pure substances combine
- Example: Sugar + water = mixture
- Sprout salad: Not a scientific mixture
(components are already mixtures)
Types of Mixtures by Physical State
1. Solid
2. Liquid
3. Gas
Unifrom and non- uniform mixtures
| S.No. | Mixture-Type | Examples | Uniform or Non-Uniform |
|---|---|---|---|
| 1 | Gas + Gas | Air | Uniform |
| 2 | Gas + Liquid | Carbon particles in the air | Uniform |
| 3 | Solid + Gas | Carbon particles in the air | Non-uniform |
| 4 | Liquid + Liquid | Vinegar (acetic acid + water) | Uniform |
| Oil + water | Non-uniform | ||
| 5 | Solid + Liquid | Seawater | Uniform |
| Sand + water | Non-uniform | ||
| 6 | Solid + Solid | Alloys | Uniform |
| Baking powder | Non-uniform |
Uniform vs Non-Uniform
Uniform:
evenly mixed
no visible separation
(salt water, air)
Non-uniform:
uneven
visible separation
(oil–water, sand–water)
Purpose of Separating Mixtures
Everyday:
get useful part, discard rest (tea, rice)
Science:
obtain pure substances
Method:
based on property differences
(size, solubility, density, magnetism)
What Are Pure Substances?
Common meaning: “Pure” = no added impurities or low-quality substances.
Scientific meaning: contains only one type of particle
Scientific definition:
A pure substance contains only one type of particle and cannot be separated into other kinds of matter by any physical process.
Key Properties of Pure Substances
- Made of identical particles
- Have fixed composition and definite physical/chemical properties
- Cannot be separated by physical methods (filtration, evaporation, magnetism).
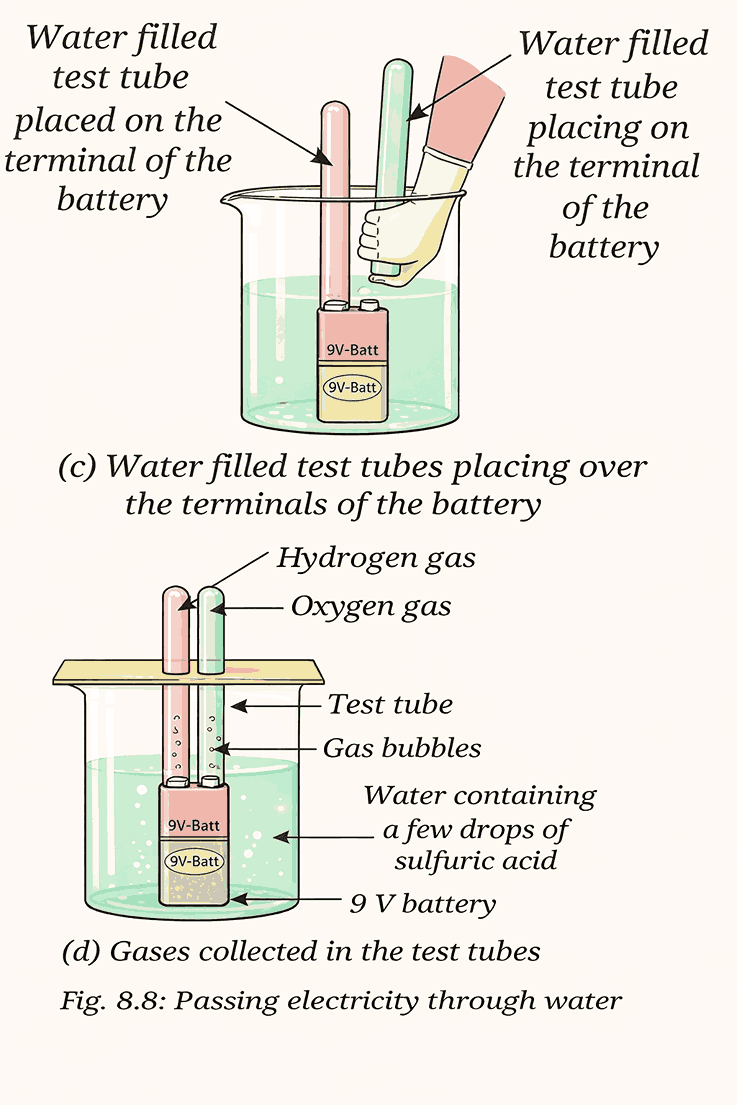
Water Electrolysis:
Proof Water Is a Compound
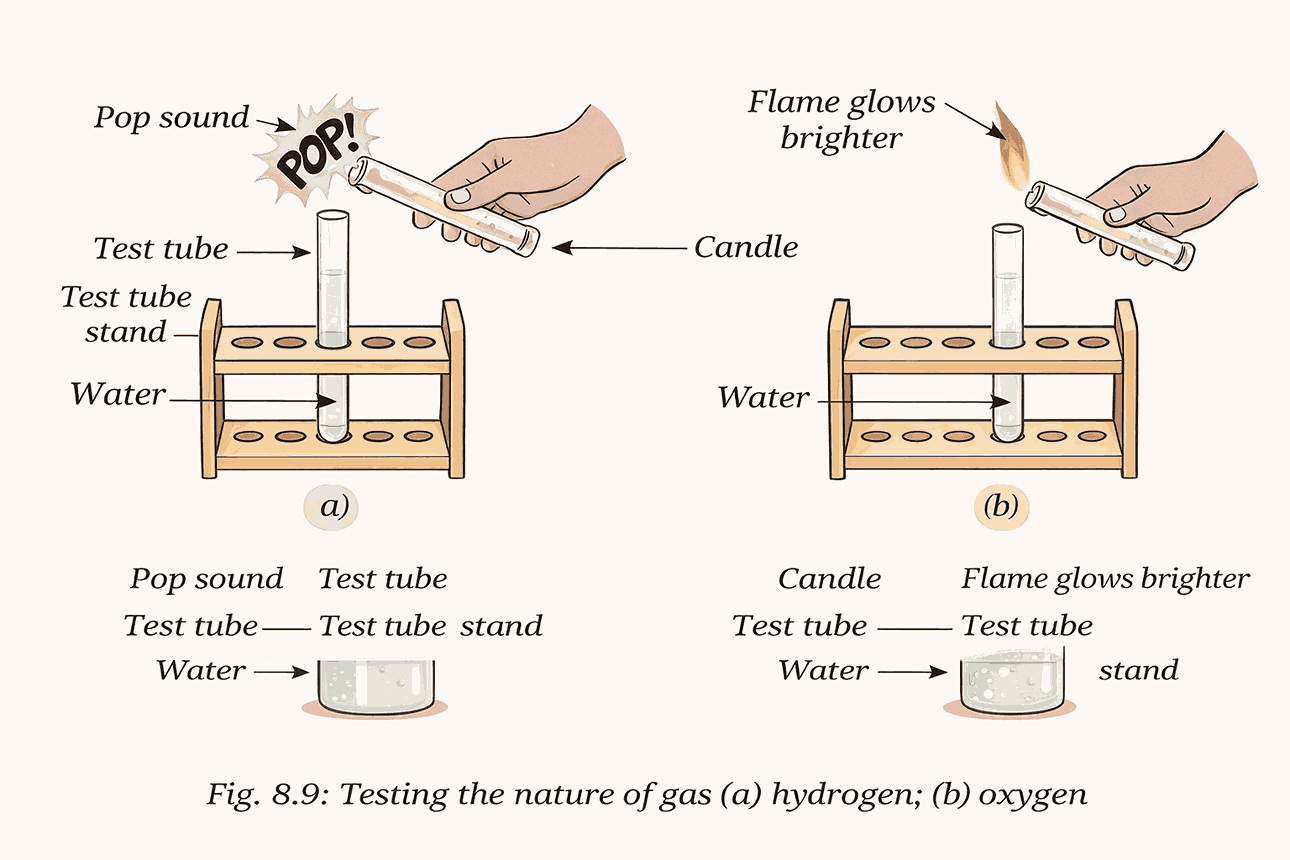
- Passing electricity through water produces two gases:
- Hydrogen: Burns with a sharp pop sound
- Oxygen: Makes a candle flame glow brighter
- Word equation: Water → Hydrogen + Oxygen
Conclusion:
Water is not an element; it is made of hydrogen and oxygen chemically combined
Elements
Definition
Pure substances that cannot be broken down into simpler substances by any chemical or physical process
- Composed of identical atoms
- Examples: Gold, silver, iron, magnesium, carbon, sulfur, hydrogen, oxygen
Classification of Elements
| Type | Key Properties | Examples |
|---|---|---|
| Metals | Shiny, malleable, ductile, good conductors of heat/electricity | Iron, aluminium, copper, gold, silver |
| Non-metals | Dull, brittle, poor conductors, often gases or powders | Carbon, sulfur, hydrogen, oxygen |
| Metalloids | Properties intermediate between metals and non-metals | Silicon, boron |
Compounds
Definition:
Pure substances are formed when two or more elements combine chemically in a fixed ratio by mass
- Properties are completely different from the constituent elements
- Cannot be separated by physical methods; need chemical reactions to break bonds
Common Compounds at a Glance
| Compound | Constituent Elements | Fixed Atomic Ratio | Notable Fact |
|---|---|---|---|
| Water | Hydrogen, Oxygen | H:O = 2:1 | Splits into H₂ and O₂ by electrolysis |
| Sodium chloride | Sodium, Chlorine | Na:Cl = 1:1 | Table salt; safe despite reactive elements |
| Sugar | Carbon, Hydrogen, Oxygen | Fixed | Table salt is safe despite reactive elements |
Mixture vs Compound: Iron and Sulfur Activity
| Feature | Sample A: Iron + Sulfur (Mixture) | Sample B: Iron sulfide (Compound) |
|---|---|---|
| Appearance | Visible black (iron) and yellow (sulfur) particles | Uniform black mass throughout |
| Magnet test | Iron filings attracted; sulfur unaffected | No attraction to magnet |
| Reaction with dilute HCl | Iron → Hydrogen gas (pop sound); sulfur unchanged | Produces hydrogen sulfide (rotten egg smell) |
| Separation | Components separable by magnet or physical means | Cannot be separated by physical methods |
| Properties | Each component retains its original properties | Iron filings attracted; sulfur was unaffected |
How Do We Use Elements, Compounds, and Mixtures?
Real-World Applications
| Stronger, more durable materials like stainless steel | Use of Elements/Compounds/Mixtures | Outcome |
|---|---|---|
| Medicine | Chemists combine elements to form new compounds | Life-saving drugs, vaccines, treatments |
| Agriculture | Knowledge of compounds used to make fertilisers | Higher crop yields, food security |
| Engineering | Design of alloys and composite mixtures | Stronger, more durable materials like stainless steel |
| Construction | Use of mixtures like wood, steel, concrete | Safe, long-lasting buildings and infrastructure |
What Are Minerals?
Minerals
naturally occurring substances with definite composition
Rocks
mixtures of minerals (visible, magnifier, or microscope)
Types of Minerals
| Type | Description | Examples |
|---|---|---|
| Native minerals | Pure elements (not compounds) | Metals: Gold, silver, copper Non-metals: Sulfur, carbon |
| Compound minerals | Made of two or more elements chemically combined | Quartz, calcite, mica, pyroxene, olivine, talc |
Everyday Uses of Minerals
- Cement: Made from calcite, quartz, alumina, and iron oxide
- Talcum powder: Made from mineral talc
- Metals: Extracted from mineral ores for construction, electronics, and vehicles
- Building materials: Sand, gravel, limestone derived from minerals
Matter vs Non-Matter
- Matter: Has mass and occupies space
- Includes: Elements, compounds, mixtures, minerals, rocks, water, air
- Not matter: No mass, does not occupy space
- Includes: Light, heat, electricity, thoughts, emotions
FAQs Nature of Matter Elements Compounds And Mixture Notes
- What is matter in simple words?
Matter is anything that has mass and occupies space. - What are elements, compounds, and mixtures?
Elements are pure substances, compounds are chemically combined elements, and mixtures are physical combinations of substances. - What is the difference between a pure substance and a mixture?
Pure substances have one type of particle; mixtures have two or more substances mixed physically. - Is air a mixture or a pure substance?
Air is a mixture of gases like nitrogen, oxygen, and carbon dioxide. - What is a mixture with examples?
A mixture is a combination of substances where each retains its properties (e.g., salt + water). - Can mixtures be separated? How?
Yes, using physical methods like filtration, evaporation, or magnetism.




