Stuck in the numerous concepts and reactions in the chapter Carbon and its compounds?
No worries, Carbon And Its Compounds Short Notes class 10 is the solution to your problems.
All the concepts and the associated chemical reactions are presented here in the short notes in clear and logical manner.
This lets you learn and memorize the concepts in a easy way.
Only the important points for the examination point of view are coverd. So if you want to understnad the concrpts then turn to your NCERT textbook.
Introduction to Carbon and Its Compounds
Why Carbon Is Important
- Carbon is a versatile element.
- Found in:
- Food, clothes, medicines, fuels.
- Paper, plastics, fabrics.
- All living organisms (life is carbon-based)
Bonding in Carbon: Covalent Bond
Why Carbon Forms Covalent Bonds
- Carbon has 4 valence electrons (tetravalent).
- To become stable, carbon:
- Cannot lose 4 electrons → requires too much energy.
- Cannot gain 4 electrons → nucleus cannot hold 10 electrons.
- So carbon shares electrons to complete its octet → forms covalent bonds.
Properties of Covalent Compounds
- Have low melting & boiling points.
- Do not conduct electricity → no ions present.
- Have weak intermolecular forces, but…
- Strong bonds within molecules.
Short Answer Questions
Q1. Why can’t carbon form C⁴⁺ or C⁴⁻ ions?
Answer:
- Losing 4 electrons needs very high energy.
- Gaining 4 electrons makes 10 electrons, which the nucleus cannot hold.
Hence, carbon shares electrons instead.
Q2. Why are covalent compounds bad conductors of electricity?
Answer:
- They do not form ions.
- No charged particles → no electricity flow.
| Keyword | Meaning |
|---|---|
| Tetravalency | Carbon has 4 valence electrons |
| Covalent bond | Bond formed by sharing electrons |
| Intermolecular forces | Weak forces between molecules |
| Valence electrons | Electrons in outermost shell |
What Are Allotropes?
Allotropes are different physical forms of the same element.
Carbon has three major allotropes:
1. Diamond
- Each carbon atom is bonded to 4 other carbon atoms.
- Forms a rigid 3D structure.
- Hardest natural substance.
- Non-conductor of electricity (no free electrons).
- Used in:
- Cutting tools
- Jewellery
2. Graphite
- Each carbon atom bonded to 3 other carbon atoms.
- Forms hexagonal sheets (layers).
- Layers slide → soft and slippery.
- Good conductor of electricity (free electrons).
- Used in:
- Pencils
- Lubricants
- Electrodes
3. Fullerenes
- Carbon atoms arranged in spherical cages.
- Example: C₆₀ Buckminsterfullerene
→ Shape looks like a football - Each carbon bonded to 3 others.
- Used in:
- Nanotechnology
- Drug delivery
Short Answer Questions — Allotropes
Q1. Why is diamond hard but graphite soft?
Answer:
- Diamond has a rigid 3D network.
- Graphite has layers that slide over each other, making it soft.
Q2. Why does graphite conduct electricity but diamond does not?
Answer:
- Graphite has delocalized electrons, so it conducts.
- Diamond has no free electrons, so it does not.
| Keyword | Meaning |
|---|---|
| Allotrope | Different physical forms of the same element |
| C–C bond | Covalent bond between carbon atoms |
| Buckminsterfullerene | C₆₀ molecule shaped like a football |
| Hexagonal array | Six-sided ring arrangement in graphite |
Versatile Nature Of Carbon
Why Carbon Forms So Many Compounds?
Carbon forms so many compounds because :
Tetravalency
- Carbon has 4 valence electrons.
- Forms strong covalent bonds with:
- H, O, N, Cl, S, etc.
- Allows formation of stable molecules.
Catenation
- Ability to form long chains of carbon atoms.
- Chain types:
- Straight
- Branched
- Rings
- Can include:
- Single bonds
- Double bonds
- Triple bonds
This property is unique to carbon.
Short Answer Questions — Versatile Carbon
Q1. What is catenation?
Answer:
Self-linking of carbon atoms to form long chains and rings.
Q2. Why does carbon form strong covalent bonds?
Answer:
Because of its small size → nucleus holds shared electrons tightly.
| Keyword | Meaning |
|---|---|
| Catenation | Self-linking of carbon atoms |
| Tetravalency | Having four valence electrons |
| Unsaturated hydrocarbons | Compounds with double or triple bonds |
| Saturated hydrocarbons | Compounds with only single bonds |
Saturated & Unsaturated Carbon Compounds
Saturated Compounds (Single Bonds Only)
Examples:
- Methane (CH₄)
- Ethane (C₂H₆)
- Propane (C₃H₈)
Properties:
- Less reactive
- Burn with clean flame
Unsaturated Compounds (Double/Triple Bonds)
Examples:
- Ethene (C₂H₄) → double bond
- Ethyne (C₂H₂) → triple bond
Properties:
- More reactive
- Burn with yellow, sooty flame
Example Structures
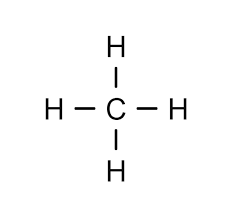
Methane (CH₄) – Saturated
Carbon shares 4 single bonds.
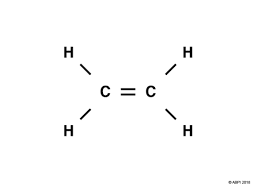
Ethene (C₂H₄) – Unsaturated
Contains 1 double bond.
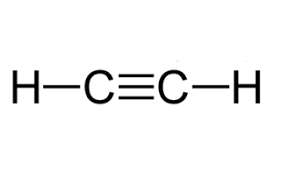
Ethyne (C₂H₂) – Unsaturated
Contains 1 triple bond.
Short Answer Questions — Saturated & Unsaturated
Q1. What is the difference between saturated and unsaturated hydrocarbons?
Answer:
- Saturated → single bonds only
- Unsaturated → contains double or triple bonds
Q2. Which burns with a cleaner flame — saturated or unsaturated?
Answer:
Saturated hydrocarbons burn with a clean blue flame.
| Keyword | Meaning |
|---|---|
| Alkane | Saturated hydrocarbon (single bonds) |
| Alkene | Unsaturated hydrocarbon (double bond) |
| Alkyne | Unsaturated hydrocarbon (triple bond) |
| Hydrocarbon | Compound of carbon and hydrogen only |
Chains, Branches & Rings
Carbon Chains
Carbon atoms bond with each other to form:
1. Straight Chains
- Continuous chain of carbon atoms
Example:- Butane (C₄H₁₀)
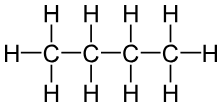
2. Branched Chains
- Main carbon chain with one or more side chains
Example:- Isobutane (another form of C₄H₁₀)
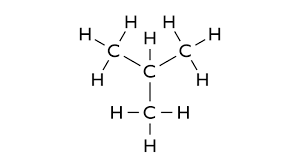
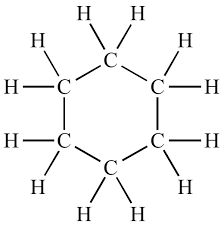
3. Rings (Cyclic Structures)
- Carbon atoms arranged in a loop
Examples:- Cyclohexane (C₆H₁₂)
- Benzene (C₆H₆)
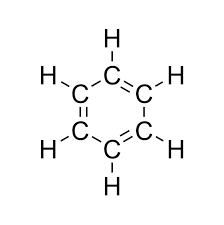
Structural Isomerism
- Compounds with:
✔ Same molecular formula
✔ Different structural arrangement - Example:
- C₄H₁₀ has two isomers:
- butane (straight)
- isobutane (branched)
- C₄H₁₀ has two isomers:
Short Answer Questions
Q1. What are structural isomers?
Ans: Compounds that have the same molecular formula but different structural arrangements.
Q2. What is a cyclic carbon compound?
Ans: A compound in which carbon atoms form a ring structure.
| Keyword | Meaning |
|---|---|
| Chain | Linear sequence of carbon atoms |
| Branch | Side group attached to the main chain |
| Structural isomer | Same formula, different structure |
| Cyclic compound | Carbon atoms arranged in a ring |
Functional Groups
What is a Functional Group?
A functional group is an atom or group of atoms that gives a carbon compound its chemical properties.
Important Functional Groups
| Functional Group | Formula | Class |
|---|---|---|
| Alcohol | –OH | Alcohols |
| Aldehyde | –CHO | Aldehydes |
| Ketone | >C=O | Ketones |
| Carboxylic Acid | –COOH | Carboxylic acids |
| Halo (Chloro/Bromo) | –Cl / –Br | Haloalkanes |
Functional groups replace one or more hydrogens in a hydrocarbon chain.
Short Answer Questions
Q1. What is a functional group?
Ans: An atom or group of atoms that determines the chemical properties of a carbon compound.
Q2. What functional group is present in alcohols?
Ans: –OH (hydroxyl group).
| Keyword | Meaning |
|---|---|
| Functional group | Reactive part of molecule |
| Hydroxyl group | –OH group |
| Carboxyl group | –COOH group |
| Aldehyde group | –CHO group |
Homologous Series
What Is a Homologous Series?
A group of organic compounds with:
- Same functional group
- Same general formula
- Similar chemical properties
- Successive members differ by –CH₂– (14 u)
Examples
Alkanes
CH₄, C₂H₆, C₃H₈, C₄H₁₀
General formula: CₙH₂ₙ₊₂
Alcohols
CH₃OH, C₂H₅OH, C₃H₇OH
General formula: CₙH₂ₙ₊₁OH
Characteristics
- Gradual change in physical properties:
- Melting/boiling points increase with molecular mass.
- Chemical properties stay similar:
- Because functional group is the same.
Short Answer Questions
Q1. What is the common difference between members of a homologous series?
Ans: A –CH₂– unit.
Q2. Why do homologous series members show similar chemical properties?
Ans: Because they have the same functional group.
| Keyword | Meaning |
|---|---|
| Homologous series | Family of compounds with same functional group |
| General formula | Formula pattern for a series |
| CH₂ group | Difference between successive members |
Nomenclature Of Carbon Compounds
Rules for Naming Carbon Compounds (IUPAC)
- Select longest carbon chain
→ This gives the base name (meth, eth, prop, but, pent, …) - Identify functional group
- Replace last part of chain name with suffix
Examples:- Alcohol → –ol
- Aldehyde → –al
- Ketone → –one
- Carboxylic acid → –oic acid
- Replace last part of chain name with suffix
- Number the chain
- Give lowest possible number to the functional group.
- Combine
- Prefix (if needed) + Root word + Suffix
Examples
1. CH₃–CH₂–OH
- 2 carbon atoms → “eth”
- Alcohol → “ol”
- Name = ethanol
2. CH₃–CH₂–COOH
- 3 carbons → “prop”
- Carboxylic acid → “oic acid”
- Name = propanoic acid
Short Answer Questions
Q1. What suffix is used for ketones?
Ans: –one
Q2. Write the IUPAC name of CH₃–CHO.
Ans: Ethanal
| Keyword | Meaning |
|---|---|
| IUPAC | Standard naming system |
| Prefix | Part added before name |
| Suffix | Part added after root |
| Root word | Shows number of carbon atoms |
Chemical Properties Of Carbon Compunds
(Combustion, Oxidation, Addition, Substitution)
COMBUSTION
What is Combustion?
When carbon or its compounds burn in oxygen, they form CO₂, heat, and light.
Examples:
- C + O₂ → CO₂ + heat
- CH₄ + O₂ → CO₂ + H₂O + heat
- Ethanol also burns with a flame.
Flame Behavior
- Saturated compounds → clean blue flame
- Unsaturated compounds → yellow sooty flame
- Incomplete combustion (less oxygen) → black soot on utensils
Why soot deposits form on vessels
Blocked air inlets → insufficient oxygen → incomplete burning → soot.
Short Questions — Combustion
Q1. Why do unsaturated hydrocarbons burn with a yellow flame?
Ans: Because they undergo incomplete combustion producing carbon particles (soot).
Q2. Why does a vessel turn black on the bottom?
Ans: Due to incomplete combustion causing soot deposition.
| Keyword | Meaning |
|---|---|
| Combustion | Burning in oxygen |
| Soot | Carbon particles from incomplete burning |
| Oxides of nitrogen/sulphur | Pollutants from burning fuels |
OXIDATION
What is Oxidation?
Addition of oxygen or removal of hydrogen.
Examples:
- Ethanol → Ethanoic acid (by alkaline KMnO₄ or acidified K₂Cr₂O₇)
Oxidising Agents
- Alkaline KMnO₄ (purple)
- Acidified K₂Cr₂O₇ (orange)
These convert alcohols → acids.
Short Questions — Oxidation
Q1. Why is conversion of ethanol to ethanoic acid called oxidation?
Ans: Because oxygen is added to ethanol.
Q2. Name two oxidising agents.
Ans: Alkaline KMnO₄ and acidified K₂Cr₂O₇.
| Keyword | Meaning |
|---|---|
| Oxidation | Addition of oxygen |
| Oxidising agent | Substance that adds oxygen |
| Ethanol → Ethanoic acid | Example of oxidation |
ADDITION REACTION
What Are Addition Reactions?
These occur only in unsaturated hydrocarbons (alkenes/alkynes).
Example:
Ethene + H₂ → Ethane
(in presence of Ni/Pd catalyst)
This is used in:
- Hydrogenation of vegetable oils → solid fats.
Q1. Which type of hydrocarbons undergo addition reactions?
Ans: Unsaturated hydrocarbons (with double/triple bonds).
Q2. What is hydrogenation?
Ans: Adding hydrogen to unsaturated oils to form saturated fats (using Ni catalyst).
| Keyword | Meaning |
|---|---|
| Addition reaction | Adding atoms across double/triple bond |
| Hydrogenation | Converting oils to fats |
| Catalyst | Substance speeding up a reaction |
SUBSTITUTION REACTION
What is Substitution?
One atom replaces another atom in a saturated compound.
Example:
CH₄ + Cl₂ → CH₃Cl + HCl
(in the presence of sunlight)
- Occurs in alkanes
- Chlorine replaces H one by one
Short Questions — Substitution
Q1. What reaction do alkanes generally undergo?
Ans: Substitution reactions.
Q2. Why is sunlight needed for substitution?
Ans: It provides energy to break Cl₂ molecules into reactive atoms.
| Keyword | Meaning |
|---|---|
| Substitution | One atom replaces another |
| Alkyl halide | Product formed (e.g., CH₃Cl) |
| Photochemical reaction | Reaction using light |
Ethanol
Properties of Ethanol
- Liquid at room temperature
- Soluble in water in all proportions
- Used in:
- Alcoholic drinks
- Cough syrups, tincture iodine (good solvent)
- Fuel (blended with petrol)
- Drinking absolute alcohol (100% ethanol) is fatal
- Long-term use damages liver and nervous system
Reactions of Ethanol
Reaction with Sodium
2Na + 2C₂H₅OH → 2C₂H₅ONa + H₂↑
- Produces hydrogen gas
- Forms sodium ethoxide
- Similar to the reaction of Na with water or acids
Dehydration of Ethanol
- Ethanol heated with concentrated H₂SO₄ at 443 K → Ethene + Water
Reaction:
C₂H₅OH → C₂H₄ + H₂O
- Sulphuric acid acts as a dehydrating agent
Ethanol as a Fuel
- Burns cleanly, producing only CO₂ + H₂O
- Added to petrol (gasohol) for cleaner energy
📝 Short Questions — Ethanol
Q1. What happens when ethanol reacts with sodium?
Ans: Sodium ethoxide and hydrogen gas are formed.
Q2. Why is concentrated H₂SO₄ called a dehydrating agent?
Ans: Because it removes water molecules from ethanol to form ethene.
| Keyword | Meaning |
|---|---|
| Sodium ethoxide | Product of ethanol + sodium |
| Dehydration | Removal of water |
| Denatured alcohol | Ethanol mixed with poisons to prevent drinking |
| Absolute alcohol | 100% pure ethanol |
Ethanoic Acid (Acetic Acid)
Properties of Ethanoic Acid
- Commonly called acetic acid
- 5–8% solution = vinegar
- Melting point = 290 K → freezes in winter → “glacial acetic acid”
- Weak acid (partially ionised)
Reactions of Ethanoic Acid
Reaction with Ethanol → Ester (Esterification)
CH₃COOH + C₂H₅OH → CH₃COOC₂H₅ + H₂O
- Pleasant-smelling esters
- Used in perfumes, flavourings
- Reaction occurs in presence of conc. H₂SO₄ (catalyst)
Reverse reaction (with NaOH):
Ester → Sodium acetate + Alcohol
(This is saponification)
Reaction with Bases
CH₃COOH + NaOH → CH₃COONa + H₂O
- Forms sodium acetate
Reaction with Carbonates
2CH₃COOH + Na₂CO₃ → 2CH₃COONa + H₂O + CO₂↑
- Produces brisk effervescence (CO₂)
Reaction with Hydrogen Carbonates
CH₃COOH + NaHCO₃ → CH₃COONa + H₂O + CO₂↑
- Limewater test confirms CO₂
Short Questions — Ethanoic Acid
Q1. How will you distinguish between alcohol and acid?
Ans: Add NaHCO₃:
- Acid → CO₂ gas forms
- Alcohol → no reaction
Q2. What is esterification?
Ans: Reaction of alcohol + carboxylic acid forming ester + water.
| Keyword | Meaning |
|---|---|
| Glacial acetic acid | Pure ethanoic acid |
| Ester | Sweet-smelling compound |
| Saponification | Breaking ester using base |
| Hydrogen carbonate | NaHCO₃ |
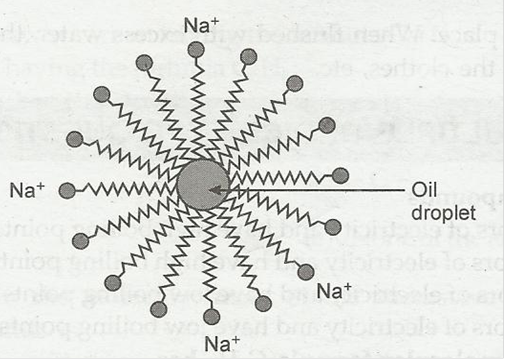
Soaps & Detergents (Micelles)
What Is Soap?
- Sodium/potassium salts of long-chain fatty acids
- Soap molecule has:
- Hydrophobic tail (repels water; attracts oil)
- Hydrophilic head (attracts water)
Micelle Formation
When soap is added to water:
- Hydrophobic tails surround oil/dirt
- Hydrophilic heads stay in water
- Forms spherical clusters called micelles
- Dirt is trapped inside micelles
- Micelles wash away with water
Detergents
- Ammonium or sulphonate salts
- Work even in hard water
- Do NOT form scum
Hard Water vs Soap
- Hard water contains Ca²⁺, Mg²⁺
- Soap + hard water → insoluble scum
- Detergents do NOT form scum → preferred in washing machines
Short Questions — Soaps & Detergents
Q1. Why do soaps not work well in hard water?
Ans: Because they form insoluble scum with Ca²⁺ and Mg²⁺ ions.
Q2. What is a micelle?
Ans: A spherical cluster of soap molecules with the hydrophobic tails inside and hydrophilic heads outside.
Q3. Why are detergents better in hard water?
Ans: They do not form scum and remain soluble.
| Keyword | Meaning |
|---|---|
| Micelle | Soap cluster removing dirt |
| Hydrophilic | Water-attracting |
| Hydrophobic | Water-repelling |
| Scum | Insoluble residue formed with soap in hard water |
Conclusion : Carbon And Its Compounds Short Notes
Congratulations on coming so far! We hope our short notes have been a helpful part of your CBSE exam preparation.
Keep revisiting these notes again and again. If you read them with concentration 20 to 30 times, you’ll find the entire chapter will naturally commit to memory.
If you found the short notes helpful, then please share it with your friends and peers.




